
Foreword
Preface
Acknowledgements
Introduction to The Haworthia Handbook – 1979 Version
Introduction to The new Haworthia Handbook – 1982 Version
Introduction to Haworthia Revisited – 1999 Version
Historical Sketch
Collectors and Contributors
Cultivation, Propagation and Plant Health
Contribution by S.A. Hammer
The Genus and Species Concept
Synopsis of Taxonomic Changes
Key to the Subgenera
Haworthia Revisited: A Revision of the Genus. (out of print)
Umdaus Press, Hatfield, South Africa, 1999
ISBN-10: 1919766081
Taxonomic revision of the genus. Comprehensively illustrated with photographs of both habitat and cultivation. Hardcover. 250 pages 21 x 26 cm.
Foreword
In 1977 Dr D.R. Hunt, formerly of Kew Gardens, London, wrote in the Cactus and Succulent Journal of Great Britain that M.B. Bayer “has emerged in recent years as the leading researcher on Haworthia“. Since the publication of the Haworthia Handbook, now a rare item of Africana, many more books and a vast number of articles have been written on Haworthia, with the popular journal Haworthiad leading much discussion on issues of topical interest. Twenty years later it is without exaggeration to say that Bayer remains the leading expert on Haworthia. His experience of these plants in the field and in cultivation is without parallel and it is consequently a great privilege for me to introduce his new and revised classification of Haworthia.
The eighteen years that have elapsed since the New Haworthia Handbook appeared has been a period of unprecedented botanical exploration of southern Africa, with more and more remote areas becoming accessible and being investigated. Succulents have received their fair share of the attention. Bayer’s previous two books greatly stimulated interest in Haworthia among collectors. To some extent this has caused a “Haworthia-craze” on a smaller but similar scale to the “Aloe-craze” brought on in this country by the monumental works on Aloe by G.W Reynolds. In some cases, this had a very negative impact on plants in the wild but it has also had the effect of stimulating a new ethic of propagation from small numbers of field-collected plants. The present book will keep this interest alive. With the vastly increased success of cultivation and propagation of succulents it is hoped that the destructive collecting of large numbers of plants from their natural habitat is a thing of the past. It is my hope that this book will stimulate competent growers to new pinnacles of achievement on propagating Haworthia, rather than renewed efforts at pillaging their natural habitats.
The great increase in enthusiasm for Haworthia among collectors has brought with it plenty of controversy over names. There is now a plethora of opinions, often strongly and not always wisely held, nor necessarily based on observable facts. Bayer has steadfastly maintained that species must be related to continuity in characters and continuity in distribution of populations in the field and that any other arbitrary selection of individual plants or populations for recognition as species, without placing them properly in their geographical context, is worthless. This rational view enabled him to reduce the “species” from about 150 to 68 in 1976. Since then, the steadily accumulating body of knowledge of these plants in the field has resulted in the reduction of the number of species to 61 in this new book, including 6 new species. It is particularly noteworthy that it has been possible to fit most of the newer collections into the scheme first proposed in 1976. In the new book he sets out to justify the changes made by presenting a much increased photographic record of each taxon and basing his conclusions on material from the now reasonably extensive herbarium record. In addition he has maintained many names at varietal rank so that unusual populations are distinguished taxonomically for the use of the enthusiast. My own experience of these plants in the wild has clearly demonstrated how complex the relationships between and within the different species are.
Bruce Bayer is to be applauded for creating some order in this maze of complexity and crystallizing out of this a further, far more detailed and much expanded account of the genus. This should once more set the standards for the group for many years to come, and also provide plenty of food for thought for those who take the trouble to read carefully what he has written.
Dr P.V Bruyns
Mowbray Cape Town
Preface
This book is written for my friends and at their instigation. It is also for those who genuinely like these fascinating plants. I have been extremely fortunate to have been able to spend so much time in the field, and still there is so much to find and see. Despite this, I am confident that this book presents a very sound hypothesis and it contains many lesser and secondary postulates. It is nested within a very wide experience and knowledge of other groups of organisms. The book rests comfortably on the 1978 and 1982 works, and is a very sound foundation for further construction. It is based on a firm definition of what a ‘species’ is, rather than on a personal construct.
Discussion and comment on the issue of the names Haworthia pumila and H. margaritifera could probably form a book in its own right and I could quote a number of other examples too. At the time Scott (and Dr L.A. Codd) worked out typification and application of the name H. pumila. I said I thought they were correct, possibly for the wrong reason. The obvious thing to do seems to me to opt for conservation of the name. It was clear to me at the time that Linnaeus had used the name Aloe pumila to cover five different elements, and I think there should be an Haworthia pumila. By choosing and citing Commelin’s figure 10 to represent his concept of Aloe pumila, Burman (1768) implicity typified Aloe pumila. Haworth (1804) selected Boerhaave’s figure 131 to represent Aloe pumila and, in so doing followed Aiton (1781), who chose the same illustration to represent his Aloe arachnoidea var. pumila. Aiton had created a new illegitimate name Aloe pumiia and he was followed by Haworth and then by Duval. Scott (1985) recognised Haworthia pumiia (L.) Duval (with the support of Codd) ahead of the then fashionable H. margaritifera (L) Haw. Unfortunately Scott overlooked Burman and implicitly used the Aition illegitimate pumila (either as a new name or a Linnaean one). The fact has also been overlooked that H. margaritifera (L) Haw. is based on a Bradley figure which is indisputably H. minima. I had intended to use the name H. maxima (Haw.) Duval as the first and indisputable reference to a species everybody knows by apparently the wrong name. At this very late stage in writing the preface, I have chosen to revert to Scott and to Burman. This is to use the name H. pumila (L) Bayer as a new combination to equate H. pumila (L) Duval sensu Scott. I believe someone has decreed the Linnaean varieties to be typified in some fashion. It is more than probable that this someone is no less fallible than several authorities who have gone before, have made similar mistakes, and also stand correction.
I came to write about Haworthia because of my fascination with plants and because I saw a real deficiency there in the available literature. I have concern that my own honesty; and the accompanying difference and reservation in my writing will not be perceived and respected by readers and other writers. Socrates wrote -The living word of knowledge has a soul of which the written word is no more than an image’. King Thamus used the words ‘only a semblance of the truth’. I have tried my best to come as close to the truth as I can, and in doing this try to observe a fundamental principle of science.
Bruce Bayer
Acknowledgements
For a work of this kind, herbarium record is an absolute foundation and necessity. The work can be no better than the evidence upon which it is built, tested and tried. No opinion in a work of this kind really carries weight unless the evidence can be examined and corroborated. Therefore I owe everything to the institutions and custodians whose work it is to curate botanical specimens and literature. These are the National Herbarium: Pretoria (PRE), the Compton Herbarium (NBG) at Kirstenbosch, and the Bolus Herbarium (BOL), University of Cape Town: Dept. of Botany – and staff. These are people who care about what they do and who set high standards. I am grateful for their tolerance and patience.
Acknowledgement is also made to those who have contributed material and particularly assisted me in the collection process, including particularly Dr P.V. Bruyns and Mr J.D. Venter. Many others have assisted in one way and another and I am deeply grateful, in respect of material contribution or otherwise, to even the least of these.
This work and its completion owe a great deal to:
1. the eternal cheerful optimism and friendship of Mrs. W. Schwegmann of Sheilam Nursery, Robertson;
2. the resolute and steadfast encouragement and support of Kobus (J.D.) Venter, his integrity, and his interest in Haworthia;
3. Steven Hammer for his interest and collaboration, and for the editing which he did on the manuscript;
4. Peter Bruyns for editing and correcting the text;
5. the publisher, Umdaus Press, and specifically François Steffens for his meticulous editing and Kotie Retief for his patient co-ordination;
6. my wife Daphne, and our children, who have managed to endure and even come to share some of my absorption in another world, without asking anything in return.
Readers should please note that the plants on the photographs are denoted by JDV accession numbers. Some of these plants are from collections made by other collectors.
These include:
Dr V.B. Reddi, Dr P.V. Bruyns, J.G. Marx, C. Marais, C. Rowe, V. de Vries, E.J. van Jaarsveld, J. Louw, J. Vlok, C. Burgers, T. Visser, C. McDowell, H. Gie, J. Bouwer, P. Fourie, C. Grobbelaar, J. Meyer, G. Lombard, D. & T. Rossouw, D. Tribble, R. Kent, P. Bosch, E. Dunne, M. Parisi, D. Cumming, D. de Kok, E. Aslander, R. Kratz and E. Esterhuizen. Their cooperation and enthusiasm is greatly appreciated. The contribution of Sheilam Nursery, the Karoo Botanic Garden and Kirstenbosch Botanical Gardens are also acknowledged.
Herbarium specimens are cited according to the system of Edwards and Leistner (PRE), and mapped to the centre points of the quarter-degree blocks, except in a few cases where the centre point represented the wrong aspect or the wrong distance between collection points.
Introduction to The Haworthia Handbook – 1979 Version
Interest in Haworthia has been very largely an amateur one, and this interesting succulent genus has a large following among the ranks of collectors. Names are critical to collectors as they are indeed to any attempt to observe and record information concerning the plants. There is an extensive literature to Haworthia and over 400 different names have been used at one time or another to refer to species, varieties, etc. The present publication is an attempt to review the history and literature of the genus and present a list of names which will be of practical value in dealing with it. This is by no means a definitive work and the writer does not pretend that it is anything but a simplistic view of a complex situation. It is however based on an extensive knowledge of the plants in the field derived from the records compiled by G.G. Smith and from the writer’s personal experience. The work was in fact made possible largely by the systematic and dedicated contribution made by G.G.Smith. The illustrations are all from photographs taken by the writer of living plants mostly from the original type localities where these are known.
In many cases more than one photograph is required to adequately illustrate a species and even colour photography cannot adequately demonstrate differences between species. The illustrations thus do not reflect the real diversity in the genus, and similarly the checklist does not express this diversity adequately either. Apart from the difficulties in applying the ranks of subspecies, variety, forma consistently, there are several populations known to the writer which cannot be confidently included in any given species and thus could merit independent species status. The rank of forma has no particular significance other than to express a particular form in a species where there may in any case be a high degree of variability. Many of the names rejected in this work could easily be resuscitated at this level. An attempt has been made to use varietal rank to denote populations with some geographically associated differences, and subspecies where such a distinction is greater.
The keys to the species must be regarded only as approximate particularly in the subgenus Haworthia where many of the couplets are indeed weak. This is particularly true towards the end of the key. The nature of the available superficial characters and their variability make it virtually impossible to construct any infallible key. The nuances of form, colour and armature and changes brought about by differences in growing conditions preclude an accurate and really worthwhile key. It is believed that the photographs will serve a better purpose than the possibly misleading keys. No attempt has been made to divide the subgenera into sections for much the same reason. The subgenera Hexangulares and Robustipendunculares are small enough to handle without sections, and in the case of the former the natural divisions may fragment it excessively. In the subgenus Haworthia there are several small natural sections leaving a residue of irreconcilable species.
A proper understanding of the genus Haworthia will have to be based on anatomical and ecological grounds, and this account is presented in the knowledge that a more objective technical account is desirable. In all, 68 species are recognized together with 47 subspecies, varieties and forms.
Introduction to The new Haworthia Handbook – 1982 Version
The original title of the Haworthia Handbook should perhaps have read ‘Illustrated Index of Haworthia names’. However, the Handbook was intended to serve a specific purpose in bringing all the names and literature of previous years together. The purpose of the second edition is to present an up‑to‑date version with necessary corrections and alterations in a new format. It is based on the G.G. Smith collection from the East London Museum acquired on loan and now housed in the Compton Herbarium at Kirstenbosch. Dr M.Courtenay‑Latimer was instrumental in obtaining quite a considerable quantity of papers, drawings and photographs from G.G. Smith’s estate. The collation of all this information and herbarium records has provided an ideal opportunity to review species concepts in Haworthia, and establish where problems exist. This was followed by many field excursions and new collections, which have all tried and tested the writer’s application of names. C.L. Scott has published several further papers which have contributed some useful ideas and called for further explanation. Notable here is the application of the older names H. pumila and H. altilinea. The latter name is not accepted in this work, but the former is. A further consequence of Scott’s work is that the name tessellata is resurrected in the place of recurva, but still as a subspecies of H. venosa.
Recent experience and observation have not led to any real solutions to the problem of recognising species in Haworthia. They mostly tend to confirm the views expressed in the first edition, in the chapter on genus and species concepts. This includes the view that species intergrade across geographic barriers and vary across ecological barriers. Very few of the more widely distributed species can as yet be clearly circumscribed to establish infallible identifications. For example, H. cooperi clearly intergrades with H. bolusii. H. bolusii has small forms which are to all intents and purposes similar to H. aranea. The small forms of H. bolusii lead, with little imagination, to H. translucens ssp. tenera. Similarly H. bolusii apparently transforms into H. translucens in the mountains near the southern boundary of its distribution range.
It has long been clear that a taxonomic solution may be possible for species together in one locality, and wrong in another. Thus H. herbacea and H. reticulata grow together and hybridise northwest of Worcester, but southwest of Robertson there are populations which cannot confidently be ascribed to one or the other species. Similarly this complex of H. reticulata/herbacea intergrades into H. turgida. The intergradation unfortunately is not through a continuous series of plants, but leaps across from isolated population to isolated population, creating an irregular pattern difficult to unravel into separate species.
The relationship between H. turgida and H. retusa is possibly more complex than the above, and is perhaps a contest of small/proliferous versus large/solitary plants. In other species too, there is variation coupled with distribution, and variation coupled with habitat. In an area of broken terrain and diverse geology this will be aggravated, particularly in low rainfall areas and on skeletal rocky soils. In some cases this is easily demonstrated in the field, e.g., H. turgida north of Heidelberg, H. habdomadis west of Ladismith, H. bolusii east of Jansenville, H. cooperi at Chalumna and at Kayser’s Beach. In other cases the presence of other species, or more dramatic differences, confuse the issue. H. decipiens, H. bolusii, H. habdomadis, H. cymbiformis and H. cooperi all diffuse into the mountains of the Baviaanskloof, and the Groot and Klein Winterhoek, merging with H. translucens and presenting something of a nightmare. Dramatic vegetative differences occur in the case of H. starkiana and H. scabra which are both closely related and practically sympatric; H. marginata and H. pumila occur practically sympatrically as do glabrous and hairy forms of H. translucens ssp. tenera. H. radula and H. attenuata grow mixed together and may in fact be only forms of one species. H. magnifica can sometimes be confused with H. retusa, which may be a true or false situation, i.e., there may be plants or populations which really are intermediate and neither one nor the other species. The same applies to H. magnifica and H. mirabilis.
Field hybridisation is definitely not the serious problem it has been made out to be. The number of known field hybrids is really minimal and the writer knows only the following:
H. pumila X Astroloba muricata (= Astroworthia bicarinata)
H. pumila X H. marginata
H. minima X H. marginata
H. turgida X H. floribunda
H. graminifolia X H. arachnoidea
H. herbacea X H. reticulata
H. cymbiformis X H. angustifolia
H. scabra X H. viscosa
H. viscosa X H. longiana
H. truncata X H. arachnoidea
There are, of course, a number of difficulties such as H. fasciata fa. browniana, H. glauca var. herrei fa. armstrongii, and H. angustifolia fa. baylissii, which may possibly be the consequence of hybridisation. The low incidence of hybridisation is a situation to be expected in a relatively young genus, as it would be difficult in the first place for species to arise until some form of isolation becomes entrenched. Hybridisation is more of a secondary mechanism operating where established species meet. In Haworthia there is definitely no sound basis on which problems of variability and identification in the field can be attributed to hybridisation.
This second edition, like the first, makes no attempt to meet the need of the collector in covering all those odd names which have arisen over the years. Many clonotypes of species described by Von Poellnitz, Uitewaal, Resende and Smith are still cherished in collections. There are also many plants bearing names by Haworth, Salm‑Dyck and Baker which may even be reasonably accurately identified. Descriptions and illustrations are available in the literature and wait for another enthusiast to collate and present.
In this revised addition, three new species replace three species which cannot be further upheld, and there are several changes in the lesser ranks. Altogether 68 species are again recognised, with 47 subspecies, varieties and forms. These species are here grouped into 3 subgenera where they are arranged alphabetically and not in any phylogenetic order.
Introduction to Haworthia Revisited – 1999 Version
The Handbook was first published in 1973 and revised in 1982. It now seems timely to revise the work again, especially in view of the very extensive interest in Haworthia, and all the collecting which has been done since then. This has led to new perceptions, and the object will be to try and synthesise these in a consistent way in a new arrangement. It appears to me that much of the new material and evidence substantiates the robust nature of the old Handbook treatment, although many of the new finds cannot be accommodated comfortably in the existing nomenclatural framework.
The Handbook was originally written to ‘present a list of names which will be of practical value in dealing with it’ (i.e., communication – the history, literature and cultivation of the genus). There were a number of very substantial differences between my treatment and that of Col C.L. Scott and this is an honest attempt at reconciliation. Generally these differences will be treated in the text as they arise and there is no associated intention to be belittling in the process of doing this. Some differences are purely due to the fact that my work did not address some tricky nomenclatural problems. In some cases they are real differences of opinion and perception, and in others there does not seem to be any good explanation for gross divergence of view other than intransigence on both sides. I hope that readers will appreciate that their own perceptions as expressed in multifarious different journals, newsletters, round robins, etc. are very often also far removed from truth. These expressions are probably driven by the same misguided feelings and fervour common to us all.
I certainly erred in not recognising the differences between the Uniondale Haworthia bayeri and the westerly H. emelyae. I was aware of difference in flowering time but was so preoccupied in trying to establish H. emelyae as a valid entity (as opposed to ‘picta’ and ‘correcta’) which also took account of H. comptoniana that I just missed the obvious. The debate still continues. Which goes to show that a lot of effort is wasted in unresolved conflict, while the need for stable reference points comes second. I did come to see that there were two species present at the locality for H. comptoniana near Georgida, and David Cumming also astutely intimated that there were two species. There are a number of other instances where I really was not certain about what path to take, and the reasons will, I hope, become clearer on close reading of the chapter on species concepts – and perhaps in the main text also.
I made a number of other errors too and these are much more obvious in hindsight. A particular case is with regard to H. dekenahii Smith and H. argenteo-maculosa Smith. In reducing them first to synonomy and then to varietal rank, I unnecessarily confounded a real problem. As pointed out above, the distinction between H. retusa and H. magnifica is problematic and the two varieties mentioned are in the trouble zone. Subsequent to the Handbook I also may have generated some problems by guessing at the identity of some troublesome finds.
When I worked on Haworthia there were very few fellow collectors. Much of my effort was directed at establishing what had in fact been collected and where. I was, and still am, more comfortable with generalising than with specifying. I was sometimes quite comfortable with tentatively identifying a collection and adding a locality to indicate that I was really uncertain eg, H. emelyae var. ‘montana’ – Swartberg Mts, which is now H. vlokii. Similarly, quite a few of my collections were identified with a two-way arrow because I could not be sure to which of two species the plants could belong. Since then the quality and extent of collecting has indeed been remarkable, although it is probably only P.V. Bruyns who has followed the proper route of herbarium documentation. J.D. Venter’s is the next best properly documented collection which, while frequently lacking the formality of the herbarium record (for which lack of herbarium space is the main reason) will at least ensure that information is preserved for posterity. These additional collections have helped a great deal in clarifying some points.
However, it appears to me that identification is always going to be a problem in Haworthia, and a sad truth that a workable key for Haworthia is an unattainable ideal. Taxonomy of succulent plants is often criticised and the amateurs who indulge are scorned for their efforts. However, there are probably many great taxonomic works by revered botanists which never receive a fraction of the critical examination that the popular genera do.
There has been a large degree of excessive, illegal, and disconcertingly self-interested collecting which has made and will make no contribution to the scientific and herbarium record. Whatever growers and collectors have to say about nomenclature and much of the necessity and nonsense that goes with it, this is what provides the reference framework for all their communication.
I have to pretend to have some knowledge of taxonomy and deep insights into genetics and speciation. These I do not have. My understanding is based on my life experience and is limited by my intellect, so it would be naive to think that everyone will agree with my perceptions. It has been disappointing to me that people better equipped than myself have often not come nearly up to my expectations. There seem to be other factors than training, education and intellect which determine the arrival at mutual and then good understanding and truth. Many decisions are by the nature of things subjective. It takes some effort to surrender one’s own opinions, especially when no good reason appears to substantiate the alternative decision. This kind of difference is illustrated in the description of H. reddii, where the population was well-known to me and referred to in the Handbook.
The Haworthia Society based in England first published its Newsletter in 1986, and this has continued as Haworthiad. This small magazine has done much to recapture the interest in Haworthia and bring it to the level it appears to have enjoyed during the years preceding World War II. My wish is to see the interest and skills of my collaborators combined as a tribute to a delightful group of plants, for whatever reason they attract the attention and interest that they do.
The list of natural hybrids can be extended to include:
H. sordida X H. woolleyi
H. blackburniae X H. arachnoidea
H. viscosa X H. fasciata
H. viscosa X H. glauca var. herrei
H. monticola X H. mucronata var. inconfluens
Many commentators in the general literature on Haworthia have since commented on the ease of hybridization across even the sub-genera. More important to me are those rare discerning commentators who have used this field to substantiate that the three groups are real and not just a product of Uitewaal’s or my own wishful thinking.
In this newer revised addition, 6 new species names appear while 13 are no longer upheld, and there are many changes in the lesser ranks. Altogether 61 species are recognized, with 105 lesser ranks. These species are again grouped into the 3 subgenera, arranged alphabetically within the subgenera.
Historical Sketch
1982 – The first written record concerning Haworthia appears to be that of Oldenland, superintendent of the Dutch East India Company garden at the Cape Colony in 1695. This record includes a list of 28 ‘aloe’ species and is found in Valentyn’s Beschrywinge van der Kaap der Goede Hoop (1726). These descriptions appear verbatim in Commelin’s Praeludia Botanica (1703) with amplified descriptions, and also illustrated. Four of Oldenland’s ‘aloes’ are now placed in Haworthia; these are:
’23 … Aloe africana arachnoidea’ (Commelin, fig. 27) now Haworthia arachnoidea (L.) Duval, although this name has not been previously associated with any field population. It is considered that the name is linked with H. setata Haw.
’25 … Aloe africana folio non nihil reflexo, floribus ex albo et rubro variegatis’ (Commelin, fig. 29) now H. venosa (Lam.) Haw. despite mention of white and red variegated flowers.
’26 … Aloe africana folio in summitate triangulari, rigidissimo marginibus albicantibus’ (Commelin, fig. 30) now H. marginata (Lam.) Stearn.
’27 . .. Aloe africana erecta, triangularis et triangulari folio viscosa’ (Commelin, fig. 31) now H. viscosa (L.) Haw. Commelin, in an earlier work of his, Horti medici Amstelodamensis (1701) described and illustrated four other Haworthia. These are:
‘Aloe africana brevissimo crassissimoque folio flore subviride’ (fig. 6) now taken to be H. retusa (L.) Duv.
‘Aloe africana folio glabro et rigidissimo flore subviride’ (fig. 9) which is perhaps best regarded as H. marginata also.
The two descriptions:
‘Aloe africana folio in summitate triangulari margaritifera flore subviride’ (fig. 10) and
‘Aloe africana margaritifera minor’ (fig. 11)
– are here both regarded as H. pumila (L.) Duval. Commelin distinguished the latter by the presence of tubercles on both leaf surfaces and being half the size of the former. These differences are not incompatible with known variability in H. pumila. The differences in the taste of the sap of the two elements is inexplicable as no real difference can be readily detected in the sap of H. pumila and H. minima (Ait.) Haw., as the only species which can be considered in this context. The statement that the two elements grow together suggests that but one species is involved.
Reynolds’ (1950) conclusion that Commelin used Oldenland’s Kruidboek verbatim regarding Aloe species, is thus not true for Haworthia. Neither H. retusa nor H. pumila are listed by Oldenland, and Commelin did not consider Oldenland’s n.26 to be the same as his fig. 9 (H. marginata). In the case of H. retusa and H. marginata, Commelin stated that at least the seed capsules were drawn from a book by (or compiled for) Nicolaas Witzen, Mayor of Amsterdam and Director of the Oriental Institute in about 1692.
Notable additions to this total of 6 species are in Commelin’s Praeludia Botanica (1715) where ‘ … margaritifera minima’ is taken to be the first real reference to H. minima; and Boerhaave’s ‘ … minima, atroviridis, spinis herbaceis numerosis ornata’, n.40 in his Index alter Plantarum (1720). This latter description is now interpreted as H. herbacea. These eight species were treated in various ways but without addition until De Candolle described Aloe rigida (1799) and Jaquin A. radula (1804). That so much confusion could ensue from such an elementary breakdown becomes understandable when it is realised that at least ten distinct segregates of Haworthia can be found in the Robertson Karoo, and a further eight in the South‑western Cape area alone. The Commelin and Boerhaave figures were probably regarded as a complete portrayal of the smaller Aloineae and subsequent collections identified accordingly.
The value of historical records of the early exploration of the Cape in elucidating taxonomic problems in Haworthia must be regarded as dubious. Even Reynolds, despite an elaborate account of early botanical exploration, cannot provide earlier references to Aloe microstigma Salm‑Dyck (1854) and A. speciosa Baker (1880), which are both abundant in relative close proximity to the Cape Peninsula. Conversely Reynolds could not relate the record of A. humilis (L.) Mill. by Commelin in 1701, to exploration. Both this aloe and H. viscosa (in Oldenland’s Kruidboek) occur at their nearest point to the Peninsula, at Calitzdorp and Barrydale respectively, both situated in the Little Karoo. From available accounts, the area as far east as Swellendam was well‑known by 1676, while Cruse had already travelled overland from Mossel Bay to Cape Town by 1668. If historical records are of any value, there is no good explanation for the appearance of H. mirabilis Haw. as late as 1804, or for that matter Poellnitzia rubriflora (Bolus) Uitewaal in 1920. Specimens of H. mirabilis may, of course, as already suggested, simply have been placed under H. retusa, while P. rubriflora could very well have been Oldenland’s ’28 … Aloe africana, folio parvo et in acumen rigidissimum exeunte’. The description of H. reticulata Haw. very common in the Robertson Karoo, as late as 1812 also emphasizes the disparities between chronological progress and botanical discovery. The suggestion by Scott (1970) in his description of H. springbokvlakensis, that H. mutica Haw. was probably collected by Bowie at Mossel Bay, also demonstrates the fallibility of this kind of historical evidence. A species (eco‑type?) barely distinct from the Mossel Bay element is common in the south‑western Cape, and at two localities, adjoins deeply rutted wagon transport routes between the Cape and Swellendam. The basis of Haworth’s nine new species described in 1804 is apparently to be found in the collecting by Thunberg and Masson. However, propagation from seed was practised from the inception of the Company garden at the Cape, and Miller (Gardener’s Dictionary, 1768) referred to a variety of H. herbacea raised from seed. It is thus certain that artificial hybrids were intruding on the taxonomic scene at an early stage. Willdenow described H. fasciata (Willd.) Haw. in 1811, Salm‑Dyck H. papillosa in 1817, and Haworth added another 13 species in 1819. Most of these additions by Haworth were of plants from Kew Garden, as were the six species added in 1821. In 1824 Haworth added a further six species resulting from collection by Bowie (1816‑1822), who undertook four journeys and travelled as far afield as Grahamstown eastwards, and Graaff-Reinet to the north. Bowie’s contribution extended to three more species by Haworth in 1825 and one more in 1828. Several of Haworth’s species and varieties are referable to Salm‑Dyck too, whose Monographia was published in seven parts between 1836 and 1863. No significant additions were made until Baker published some 17 new species in 1880, all stemming from collecting done by Cooper. Cooper was in South Africa from 1859 to 1862 and Reynolds briefly records that Cooper resided at Worcester before moving to Algoa Bay, inland to Graaff-Reinet and then through the Orange Free State to Natal. There does not seem to be much hope for determining the precise geographic origin of Baker’s species and hence elucidating some of the problems these involve. Baker was responsible for the revision of the whole genus in Thiselton‑Dyer’s Flora Capensis (1896). This was followed by Berger’s monograph of Aloe in Engler’s Das Pflanzenreich (1908). New species of Haworthia were published by Wright (1‑1907), Marloth (2‑1908, 1912), Schonland (2‑1910, 1912), Archibald (2‑1940, 1946), Barker (1‑1937), Resende (5‑1938, 1946), Fourcade (1‑1932), Scott (4‑1965, 1968, 1970, 1979), Hardy and Mauve (1‑1967) and Bayer (6‑1972, 1973, 1981). More extensive contributions were made by Von Poellnitz from 1929 to 1940, Smith, 1943 to 1950 and Uitewaal, 1939 to 1948. J.R. Brown published many commentaries on Haworthia and in 1957, in “A brief review of the genus Haworthia”, noted the existence of 160 species and 210 varieties! An attempt to catalogue these names was made by Jacobsen in his Handbook of Succulent Plants vol. II, published first in German in 1954 and in English in 1960. This was followed by a review of the sections and the species comprising these by the same author in Kakteen und andere Sukkulenten (1965). In the now vast and often inconsequential literature on Haworthia, mention can be made of the Haworthia Review, a roneoed publication of ‘The Succulent Liliaceae League of America‘. This first appeared in 1946, was edited by J.W. Dodson and one number was accompanied by a comprehensive check‑list. Also of passing interest are the publications by Farden and Zantner, and more recently by Fearn, Pilbeam (including booklets, The first 5O Haworthias and The second 50 Haworthias), Speechley, Scott and Bayer in publications of English, American and Australian cactus and succulent societies.
The genus has not lacked interested researchers so much as good observation of the plants, their affinities and distribution in their native habitat. It is particularly unfortunate that G.G. Smith discontinued his work at a critical stage when he perhaps could have made a major contribution. In 1947 he wrote ‘Views on the naming of Haworthia’ published in the Journal of South African Botany, in which he presented some of the difficulties arising from lack of authoritative field work. If anything this was a plea to be allowed to work to a conclusion, unhindered by duplication and developments elsewhere. Resende took violent and personal exception to Smith’s comments and replied in acrid tones in ‘Variability and taxonomy in the genus Haworthia’ in Portugaliae Acta Biologica (B) (1949). This attack undoubtedly led to Smith’s retirement from the field. Despite being responsible for several errors of the same magnitude criticised in his paper, Smith did leave an unrivalled legacy of herbarium specimens, photographs, field notes and literature compilation. Von Poellnitz was killed during the allied bombing of Berlin in 1945, when a straying bomber pilot mistook his baronial castle for an industrial target. Familiarity with his work arouses some sympathy for the errors which he made. Obviously open‑minded, with a willingness to concede misinterpretation and change, it is conceivable that he could eventually have revised the genus adequately. Most of his material was destroyed during the war, but fragments are still available in the herbarium at Berlin (see Appendix I). Flavio Resende was a highly regarded Portuguese botanist who published several papers in Portugaliae Acta Biologica, Broteria, Boletim da Sociedada Broteriana, and Memorias da Sociedada Broteriana. Nearly all his new species and varieties were of unknown or garden origin and included polyploid hybrids. His work is thus respectfully discounted in this review.
Collectors and Contributors
1976 – The role played by private collectors in adding to information on Haworthia has been a considerable one although not ever fruitfully collated. The revival of interest in the genus stems from Dr Karl von Poellnitz working at his home at Oberlodla in Germany, coupled with the collection by Hans Herre at Stellenbosch, F.R. Long at Port Elizabeth, J.R. Brown in America and Wilhelm Triebner in South West Africa. Most of the plants received by Von Poellnitz were routed through Herre, Long and Triebner. Herre’s contribution in the field of South African succulents generally, is very well known and does not need elaboration. As an enduring record, very little remains of an accessioned and labelled Haworthia collection at the Stellenbosch University Garden. The accessions are rather too sparsely recorded to be of much value in tracing localities, which are with the wisdom of hindsight, more important than then appreciated.
Triebner was a nurseryman at Windhoek ‑ an indefatigable collector and enthusiast ‑ his contribution is notoriously inaccurate and unreliable. Many collections were confused, localities and collectors also frequently being incorrect. Plants distributed by Triebner were on many occasions misnamed, although it is quite certain that Triebner was not entirely at fault. Many independent observers relate the experience of submitting plants to so many recognised authorities and receiving that number of different identifications. J.R. Brown, in correspondence with Long, noted that names of one‑third of plants sent for re‑identification did not agree with previous naming by Von Poellnitz. Long was superintendent of parks at Port Elizabeth from 1930 to 1940. Taking a keen interest in Haworthia, he built up an enormous collection which he did not administer himself. In a protracted and sporadic correspondence with Brown, he made very little contribution in the way of constructive and informative comment. In fact, apart from a compilation of photographs and descriptions, the written record of his activities over a period of 25 years is disappointing. Had it not been for Dr H.G. Fourcade, forester, surveyor and botanist, it is likely that Long’s contribution would have gone completely unrecorded. Dr Fourcade saw Long’s collection in 1938 and recognised the importance of a collection containing co‑types of nearly all Von Poellnitz’s species. With the help of Miss Eily Archibald, a check-list was prepared and Fourcade took 230 photographs of plants in Long’s collection. Six sets of photographic prints were eventually prepared and, as intended by Dr Fourcade, form a useful reference in further study of the genus. With the outbreak of the second world war, Long became involved in the construction of airfields and his plants were left in the care of Mr F.J. Stayner (later Curator of the Karoo Botanic Garden at Worcester).
After the war, the entire collection was handed to Mr G.G. Smith, engineer, businessman and naturalist at East London. Smith collected extensively and contributed many new species and varieties. Despite the vast record handed by Smith to Kirstenbosch, together with his collection in 1957, there was very little to indicate in what direction his concepts of Haworthia were developing. Several notes indicated that he had new species and varieties in mind for publication, and also a few deletions. It was only with the recent acquisition of Smith’s papers, that it was possible to see that he was contemplating a whole series of species and varieties. He was proposing 13 new species, 18 varieties and 2 subvarieties in the H. cymbiformis complex alone, while in the H. viscosa complex he contemplated 4 new species and 14 varieties. In all there were 18 new species, 65 varieties, 4 subvarieties and 1 forma in draft manuscript. Contributors to Long’s collection included nearly all the persons commemorated in Von Poellnitz’s species names. G.J. Payne of McGregor, a pianist, church‑organist and collector of sheet‑music, sent plants mostly to Triebner, and later to Long and Smith. Mr Rossouw was a policeman stationed at Napier who collected with Payne. P.L. Meiring was a speculator in land and a horticulturist, who also sent plants to Triebner, Herre and Long. Hurling and Neil were two bachelors who owned the Winton nursery at Bonnievale and who submitted many plants to Kirstenbosch, Triebner and Long, for naming. Mrs Dora van der Bijl was president of the now defunct South African Succulent Plant Society, and lived at Great Brak. Mrs E.D. Helm was also a house‑wife at Great Brak near Mossel Bay and was responsible for the discovery of H. parksiana V. Poelln. Mrs Emely Ferguson, wife of a surveyor, lived at Riversdale. Miss G.V. Britten was the curator of the Albany Museum herbarium at Grahamstown, where Miss G. Blackbeard also lived. Mr C.H.T. Woolley resided at Addo, Mrs K.E. Jones at Steytlerville, Mrs H. Blackburn at Calitzdorp, W.A. van Ryneveld at Somerset East, M. Stiemie at Kirkwood, Mrs E.B. King and her son Gordon at Port Elizabeth, Mrs E. Braun at Kendrew, W.R.B. Marais at Beaufort West, Mrs L. Lategan, Mrs G. Morris and L. Smith lived at Oudtshoorn, Miss A. de Klerk at Tulbagh and Major H. Venter collected in the South‑western Cape. Several collectors operated fairly independently and these include M. Otzen, whose localities were fortunately recorded by Dr J. Luckhoff. A.J. Joubert was a teacher at Ladismith, as was C. Beukmann who retired at Bonnievale. G. Mclaren appears to have been involved in a nursery operation with L. Leighton near Stutterheim and may have been responsible for some of the localities dubiously recorded at Grahamstown by Von Poellnitz. S. Lockwood‑Hill was for a period magistrate at Laingsburg and W.E. Armstrong, stationmaster at Adelaide. As Smith took over Long’s collection, naturally many of these persons are also involved in Smith’s personal records. Here Dr M. Courtenay‑Latimer’s contribution was certainly considerable although not recorded to any degree. J. Dekenah, photographer and naturalist at Riversdale, also helped Smith considerably and his localities are the best recorded of all. Dr Nortier of Clanwilliam sent in plants from the Western Cape as did Miss E. Esterhuysen of the Bolus Herbarium, Cape Town. Mrs A. Taute lived at Molen River between Oudtshoorn and Uniondale. Prof. R.H. Compton was, of course, Director of the National Botanic Gardens at Kirstenbosch, where H. Hall was curator of succulent plants. Mr L.A. Malherbe, owner of the Sheilam nursery at Robertson, was specifically interested in Haworthia and submitted the original specimens of H. comptoniana Smith. Captain D.R. Keith of Stegi, Swaziland, also sent plants to Smith.
Several overseas collectors built up good collections from which relatively little useful information has ever flowed. This, together with the failure of local collections to provide any permanent and reliable record, should be a clear indictment of collecting per se as a corollary of preservation and scientific record. A specimen without a locality record is a liability in taxonomy, and Brown records his disappointment at Von Poellnitz refusing to accept plants from America for certainly that reason. Any plant considered out of its natural habitat and without reference to the population from which it comes, particularly in Haworthia, has to be regarded with caution. Thus even J.R. Brown’s admirable commentaries and photographs are at most a reflection on the contemporary taxonomy and interchange between collectors, rather than a contribution to further order in the genus. J.T. Bates, tramway conductor of Hounslow, England, was in a better position as many of Von Poellnitz’s plants found their way into his collection. Whether his collection will have retained its value as a privately administered record of Von Poellnitz’s work is doubtful. Brown felt that even the Royal Botanic Gardens at Kew had failed in providing any enduring record of Baker’s work, and Von Poellnitz must have felt this too regarding Wright’s H. pearsonii. This role of a botanic garden in relation to preservation of types is a vital one where herbarium material cannot (by ordinary methods) be depended on as a certain clue to identity. However, the conservation of a species with specific and restricted habitat preferences will depend on the preservation of habitats as the only effective and realistic method. In recent times Col. C.L. Scott has published widely, often expressing opinions at great variance from those in this work. All these have been considered and incorporated wherever possible.”
1999 – A great deal should be added here to account for the huge volume of additional information that has accumulated in the informal literature, and among collectors and growers. Seed propagation and selection are far advanced. Many collectors are very knowledgeable and authoritative comment is almost the order of the day. Only one thought needs expression and repetition: a revision like this is only an hypothesis built on the experience and knowledge of the author. It stands and falls on information. This is very largely the dry and seemingly uninteresting herbarium record. However, without that tangible physical evidence, identifications become hearsay and idle talk.
There is a lot of evidence to suggest that field-collected plants are being commercially exploited. Two kinds of people are responsible – those who physically remove the plants from the field, and those who eagerly and irresponsibly buy or receive such plants. I came early to the point where I could really only justify my own aquisitiveness by going to work for an organisation whose function, it seemed to me, was to collect and conserve plants. However, I was disappointed to find that such an ideal is also too subject to human failing and organisations seldom have the motivation and skills that personal interest generate. However,by far the greatest change and losses that I have observed in the field are those directly attributable to physical damage and destruction of habitat. Nevertheless, the potential for such loss should never be construed as an excuse to remove plants from habitat that simply ensures non-survival. Cut collecting can be done in such a way that there is no detriment to natural populations. Offsets, seeds and moribund plants are often available.
There is no doubt that a work of this kind stimulates interest and leads to further collection and potential exploitation of plants. However, the positive side is awareness of those plants and their survival status that also develops. It is ridiculous to suggest that there should be no collecting when life subsists on life, and we harvest from nature in so many other ways. But we need to tread the world lightly and learn circumspection and conservatism.
Cultivation, Propagation and Plant Health
1982 – In nature, haworthias are very particular as to habitat. Where several species occur together, they very seldom share the same ecological niche. Species also change as habitats differ and so it is surprising to find how easy they are in cultivation. It is generally recognized that haworthias can be grown in nearly any potting mixture based on a loamy soil. The essential requirement is good drainage, although this is not necessarily what they always get in the field. Most species occur naturally under the protection of other plants or in rock crevices and crannies. They very seldom occur under a closed canopy of larger plants and in the field one looks for haworthias in stable habitats, in rocky situation or shallow soils. Established plants can tolerate full exposure and it is in such plants that really beautiful colouring occurs. In some species the leaf tips naturally die back and it is unusual for H. longiana for example, to have tips on the older leaves. In H. lockwoodii and H. semiviva the leaf ends die back to afford the plants a protective umbrella of dry white leaf tissue. The complete dying away of the roots that so often occurs in cultivation is quite unknown in the field. Occasional large moribund clumps may occur where the centre rosettes begin to die but this is not the same condition as that of the cultivated plant losing its roots. This root loss is due to poor drainage and a wrong watering schedule. A plant may tolerate a heavy soil if it is watered less frequently. Mortality in the field seems to be very low whereas in cultivation plants may show signs of aging and frequently die. The development of a stem in the characteristically stemless species is a bad thing. Root development occurs immediately under the lowest living leaves and the stem beneath these roots is of no value to the plant. In cultivation this superfluous stem may remain alive to the detriment of the plant and a good root system. When re-potting plants care must be taken to trim away excess stem tissue. It is then often best to remove all roots and so encourage a completely new system. It appears that nearly all species will grow from a whole leaf cutting if the leaf is taken with some remnant of stem tissue – and if the timing is right.
Haworthias are essentially winter-growing plants and all species occurring west of at least Port Elizabeth respond to a winter growth cycle. January, February and March are too hot and dry for haworthias to grow. These statements will be contested by growers in summer rainfall areas and naturally by growers in the northern hemisphere where the seasons are reversed. However, successful cultivation also requires intuitive skills and there is no doubt that different approaches will work for different people. Propagation of haworthias is more often than not by vegetative offset, and the most common species represented in collections, are represented by proliferous, adaptable clones. Not all clones of the same species respond in the same way, and certainly a species considered to be non-proliferous may have individuals which are. Cultivation is an artificial situation. There is a selective factor in favour of easy-to-grow clones. The presence of self-sown seedlings and the speed and vigour at which they grow is evidence of this. Plants which do not normally offset can be forced to do so by damaging the growing point. Alternatively the entire plant may be cut into separate pieces, or only whole leaves used for propagating purposes. Roots may be treated the same way as leaves, although there is no documented success story. Various authors have now suggested or shown (Majumdar and Sabharwal, 1870; Wessels et al.,1976; and Shargi et al., 1980) that tissue culture is a feasible proposition.
Because of the penchant to hybridise in cultivation, seed propagation has been greatly neglected. Indications are that the species are all self-sterile which means that genetically different clones must be used for cross-pollination. The parent plants must also be isolated from bird and insect pollinators. Pollination is very simply effected by inserting a fine bristle into the flower tube immediately under the upper-middle lobe. The bristle is then inserted similarly into the flowers of the second plant. Pollen is released from the youngest flowers and so pollen should be collected from the upper open flowers of the one parent and transferred to the lower unwilted flowers of the other. The quantity and quality of pollen shed is not consistent and seems to be associated with weather conditions and plant vigour. The seed capsules begin to form immediately after successful pollination and may ripen within a few weeks. Not all species and clones are equally fertile and results can sometimes be disappointing. Failure of pollination is indicated by the early abscission of the pedicels. Very little is known concerning either how soon seed should be sown after the capsules open, or viability of seed. Indications are that seed should be collected only from dry, splitting capsules and sown at least within a few months of collection. Flowering time is not a good indication of when to sow seed as it varies very much from species to species. In the strictly winter rainfall region there are species in which the seed ripens in October, December and February. No documented results are available to indicate the best sowing times. At Worcester seed has been germinated successfully at all times of the year as it becomes available after flowering. A well-drained soil mixture with some compost is used and pebbles of 3-5mm diameter are scattered over the prepared sowing surface. The seeds are lightly covered with the same soil mixture and the tray finally sprinkled with smaller pebbles. These both support the seedlings, prevent washing away of the seeds, and provide a surface mulch. Watering can be done by standing the entire seedling tray in a water bath, by fine sprayer from above, or by watering onto a piece of paper laid over the pebble surface. The tray must be kept moist for the first few days and in semi-shade. A glass covering to maintain moisture may also be used early on. The seed should begin to germinate within 7-10 days and if not up after 4 weeks can reasonably be considered a failure.
Haworthias are as a rule not particularly prone to either diseases or insect pests. The Robustipedunculares are more susceptible than other species to mealy-bug attacking the heart of the plant. In outdoor cultivation there is also a weevil which attacks the plants in the same way as a bigger version attacks aloes. The only other pest of note is a white scale-insect. All these problems are very easily controlled by physical destruction. If the infestation is already out of control resort has to be made to any of the multitude of insecticide/fungicide combination dusts available. Diseases are also seldom encountered but the softer species of the subgenus Haworthia can develop a devastating heart rot and be lost before one is aware of a problem. However, this is a problem preceded by loss of the roots. If the disease has recurred, plants must be regularly checked for firm root anchorage. Excess dying of the lower leaves is also symptomatic. The cause of the problem is a fungal disease initiated by unsuitable growing conditions such as poor drainage, poor ventilation, excess moisture or humidity and perhaps an other-wise unsuitable soil mixture. Again recourse can be made to a suitable fungicide for watering onto the plants. However, it would be better to first use a dust formulation and allow the plant to dry off. If poorly rooted then the whole plant must be cleaned of all dead and moribund tissue, dusted with a fungicidal compound and carefully replanted in fresh soil.
1999 – Contribution by S.A.Hammer
Haworthia is a diverse genus in many respects, not the least in the varied horticultural requirements of its species. Most of these can adapt to widely different styles of growing, and almost all of them are easy to grow badly. A few seem universally fussy about what they need in order to grow well. It is therefore unsatisfactory to generalize, but perhaps a few points can usefully be made here. Some of the practices I discuss here are idiosyncratic, but they do work for me and can be assessed against, or added to, other methods which also work. It is fortunate that there are now so many keen people working with the genus horticulturally.
Roots are at the heart of the matter. I sometimes tell myself that I do not grow plants, I foster root systems; and while this may be only semantic, it does encourage me constantly to consider root health. Roots are of course hidden from view, but an experienced cultivator can glance at a potted haworthia and obtain a good idea of root health.
Healthy roots anchor the plants firmly and symmetrically; they absorb large amounts of water quickly; and they do not rot, although they die off periodically and systematically. Excessive root loss is taxing, but it is true that haworthias can regenerate roots with amazing speed. Not all haworthias have the same type of roots; consider the fat, stilt-like, long-lasting roots of H. truncata versus the finer stringy roots of H. venosa.
Roots are affected by many factors: the richness, acidity, tannic content, texture and aeration of potting media; the watering regime (depth and rhythm); and heat. The latter factor is critical. If certain species need heavy shade (H. pulchella is a good example), it is not so much that their leaves cannot endure bright light as that their roots cannot function well if they dry out quickly, as happens, of course, in bright and hot situations. Some plants have trouble establishing new roots in a hot environment. For example, a repotted H. angustifolia will re-establish very well below the bench, and can then be introduced to higher light; but it will have great difficulty if introduced to strong light before root formation is secure.
Why not grow them all in the shade? In a sense that would be safer, and I have tried it, but the results are generally unattractive; leaves bloat and turn a dull camouflage green, compactness is impossible, and (with some species) rot is more likely. Even worse, the species begin to look alike! Cultivation should emphasize the individuality of plants, not mask it. So it is usually good to give haworthias as much light as they can tolerate without risking root loss, tip burn, or the desperately self-shading bunched-in look that says: help! A bright and cool ambience is ideal. Bright = light strong enough to keep lithops from looking like newly-risen toadstools; cool = under 30°C, though plants will certainly tolerate far higher temperatures. Some species will tolerate frost, but it is better to avoid temperatures below 5°C. In any case good air movement is vital.
One of the main difficulties with haworthias is that they do not readily wilt or wrinkle; they do not give one an immediate signal of their needs. This delayed response always puts one at a remove from the plant’s condition, it is like reading last year’s newspaper. It is true that a heavy watering is quickly followed by obvious swelling and glossiness, but the effects (which are often cumulative) of subtler factors – misting, light feeding, adjustment of fertilizer ratios, slight changes of position, repotting, leaf removal, and, especially, root loss – take longer to manifest themselves. A virtuoso grower, Bob Kent, once told me that last month’s sunlight = this month’s leaf colour. It took me a long time to appreciate what he meant. Colour change can be so gradual as too imperceptible to an ardent daily viewer, and it can be positive (an increase in the reddish tints which attract most of us) or negative (reddish tints are also often indicative of stress).
Haworthias burn in an obvious way only if they are suddenly introduced to a grossly brighter environment. They may, however, darken quickly if they are moved from dull quarters to a situation in which more ultraviolet is available; they may turn nearly black overnight. This is not burning, exactly, but it can compromise plants all the same. Certainly it slows them down.
As a corollary to Kent’s observation, I would add that last year’s feedings = this year’s beauty; the effects on symmetry (and on the expression of leaf patterns) are that long-lasting. Should one feed at all? That partly depends on one’s patience. But there is a sense in which feeding= strengthening. And there is no virtue in waiting four years for a starved seed1ing to mature, when a compact look can be obtained in one-quarter the time, via dilute but frequent doses of low nitrogen fertilizer! Such feeding will also improve flowering and seed-set on mature plants. It will, however, encourage offsetting, which can be a bit excessive.
Soil mixtures have caused infinite anxiety. Many growers change their mixtures every few years and find that nothing works equally well for everything. Nothing can: roots and needs are too diverse, but one can adjust proportions here and there. A light mixture (i.e. fine-textured humus plus inert drainage material like pumice, gravel, perlite, or clay aggregate) is open in texture and will require frequent wetting. A heavy mixture (i.e., loam + sand + drainage material) stays wet for a long time, which can be an advantage in a dry warm climate, a fatal liability in a damp one. People who love the act of watering, for whom watering is synonymous with plant enjoyment, should either use a light mix, or employ a hand mister frequently and avoid the hose. But it remains true that haworthias will adapt to a wide range of potting media as long as the grower adapts along with them.
Whatever the soil one uses, a top dressing of grit, gravel, or pebbles is normally beneficial and always attractive. In a hot greenhouse, dark pebbles can actually cook plant tissue, so pale ones are better; they also show off the plants more effectively. Mulches do conceal the condition of the soil, but one learns to judge that simply from the state of the leaves.
I have avoided the difficult question: how often to water; it is too individual. However, for plants in full growth, a cycle of:
1 – 2 days of saturation
3 – 4 days of even moisture
1 – 2 days of near-dryness
gives a rhythm which is effective in arid climates. In damp areas, and during the dampest periods anywhere, the cycle will be radically slower. In any case, small shallow pots dry out much faster than one imagines; deep pots, on the contrary, have hidden and dangerous reserves. Whatever the pot depth, one can consider an alteration of infrequent deep watering, in which water runs copiously out of the drainage holes, and frequent shallow watering – almost a splashing on the surface – which amounts to a light refreshment, perhaps mostly for the grower.
Too-frequent deep watering will lead to root loss and stem rot; it will kill some haworthias and turn others into green behemoths. Slightly overwatered plants tend to develop cracks or crevasses on their leaf surfaces (curiously, the upper, not the lower surface, is subject to this); such cracking is unsightly though not injurious. I might note that some clones crack even if they are watered gingerly; as a matter of good horticultural practice these should not be propagated. The syndrome is genetically based and cannot be wholly eradicated. In my experience H. magnifica var. atrofusca, H. truncata and H. emelyae var. comptoniana are particularly prone to this.
Many species grow continuously; their life is a perpetual spring. Has anyone ever killed, or even discouraged, H. cymbiformis? Others have obvious bursts of growth which should be observed and encouraged: one should run with the tide. Generally such tides flow in late winter and early to late spring. I will even water hyperactive plants twice in a row, on successive mornings, but only if the air is warm and dry.
Some Haworthia species have unmistakable dormancy syndromes, possibly because they are adapted to a winter rainfall regime: H. lockwoodii and H. semiviva are the most obvious of these. Summer is the time for their papery sleep; during this period they need only light watering, enough to prevent complete desiccation, not of the leaves, which are already desiccated, but of the roots and meristematic core. Other species turn inert and unresponsive for long erratic periods (e.g., H. bruynsii and H. springbokvlakensis, those unrelated behavioural twins). Generally this torpor occurs in winter, when light misting is sufficient to maintain a sluggish viability, but it may also occur in high summer, when extra shading is the safest response.
Pollination is now taken seriously by many growers. It might be helpful to mention that seed production is best on well-fed plants and that flowers are most receptive a day or two after anthesis (first opening). I generally pollinate a flower twice a day, morning and evening, two days in a row. I spray my hands with alcohol when shifting from one species to another to prevent accidental transfers, because the barriers between species – particularly in the subgenus Haworthia – are slight or absent.
Some species are usually difficult to pollinate. H. limifolia is a good example. Other species may elude one grower and not another; for me H. koelmaniorum is recalcitrant, for an excellent grower in torrid Arizona it makes masses of seeds, but then his plants look better and healthier than mine. He successfully uses a fine fibre of nylon as a pollinating tool; very narrow camel’s hair brushes are ideal but expensive.
Clones of most species exhibit some self-fertility but it is difficult to prove the absence of any animal vector. Often one will notice that the topmost flower of a neglected inflorescence ripens into a fat fruit; it is always worth sowing the contents. (By the way, the offspring will not necessarily be uniform.) In other cases, the whole inflorescence seems to ripen at once in a rush, skipping no fruits; this is certainly an instance of self-fertility and the offspring will usually be self-fertile as well.
Haworthias can be propagated by seeds, leaves, offsets, stolons, coring, tissue-culture, some even from roots. Seed raising is the best method for anyone who enjoys the promulgation of variability; tissue-culture is more efficient but it results in a massive sameness.
For seed work, start with good seeds! Much seed in the trade has a low endospermatic content – the seeds are well-formed but small – and the resultant weakness can cause much trouble. Seed germinates best when a few weeks old, better than when newly ripe, but it will keep for a few years, up to seven in my experience.
Seed can be sown at any time of the year. Preferably one should avoid the hottest and coldest periods unless one sows indoors under lights; indeed I sow all seeds under lights, which gives me far greater control of quasi-sterility, insects, and temperature, ca. 20 – 25°C being ideal. The lights can be the standard “cool” tubes, but those work best when paired with broad-spectrum “grow-light” tubes.
For sowing I use a commercial sowing medium (peat moss treated to improve its wettability + a little sand + vermiculite) which I cut with an equal part of fine-screened pumice (>2mm diameter) or perlite. I sterilize the mixture in the microwave and bleach the pots; this is tedious but it pays off. I fill the pots, tamp firmly, sow the seeds, and cover with a thin layer of grit. I sow the seeds in small pots, up to 100 seeds per pot. I prefer very small plastic pots (30 x 30 x 30mm); these dry out quickly, which means that I can water them frequently and thus administer frequent doses of fertilizer. It is important not to sow so thickly that seeds cover each other, leading to a tangled chaos.
I soak the sown pots in distilled water, draining them after being thoroughly saturated (an hour or so). The pots are placed ca. 15 cm under fluorescent lights which are never turned off, and are covered with tight-fitting plastic. The next day I make sure that the plastic shows a beaded film of moisture; if it does not, I mist the pots. After four days I remove the plastic, misting the pots twice a day thereafter. Germination should occur within a few more days, 6 – 14 from the time of sowing. After germination I mist the pots once a day but I avoid saturation; aeration in the soil is critical. A small fan kept on perpetually (along with the lights) helps to prevent damp-off and algal growth; the latter can also be stemmed by progressively mulching the seedlings.
I transplant seedlings at the 4 – 5 leaf stage which usually occurs for me when they are ca. two months old; the unspotted seedlings are soaked in an hour-long bath of distilled water to which I add some Vitamin B1. I use the same medium as I used for sowing (though sterilization is unnecessary and particle size can be larger). If newly transplanted seedlings dry out, they will have a hard time re-rooting, and of course the soil mix may be difficult to re-wet.
Not all species have the same rate of growth, either as cuttings or seedlings. H. bruynsii is always slow, taking at least four years to reach flowering size from seed under my conditions. H. sordida, H. scabra and H. koelmaniorum are also slow although the latter can expand quite rapidly once it passes the juvenile stage. The retuse species are much faster and most can be in flower at 12 – 18 months. H. bayeri and its ally H. truncata var. maughanii are quite slow, both pass through a tediously prolonged (and similar) juvenile stage in which they are not quite themselves.
With all species, extra shading will promote seedling growth. Though this is at the expense of colour, that can quickly be (re-)gained. One should experiment with placement of seedlings; full morning exposure and afternoon shade are the obvious poles but many gradations between these states are possible. Seedlings (and adults) are highly responsive to changes in light and many troubles can be cured or courted by a simple shifting of pots.
Most haworthias will root from leaves. Thin leaves, e.g., those of H. arachnoidea, are very difficult to root, and firm ones, e.g. H. sordida, are slow, taking many months. Plump leaves and a bit of stem should be removed from depotted plants, dusted with fungicidal rooting powder, and placed in a sowing medium in a dim corner of the greenhouse. I mist them once a day, lightly; otherwise I ignore them. They should start to callous in a few weeks and root soon thereafter, but differentiation can take months. Incidentally, leaf removal is probably the least damaging way to collect material in the field if the mother plants are not disturbed by it.
Offsets also amount to propagation from leaves, but the plant itself does the work, forming plantlets at the base of leaves which are nearing the end of their term of duty. The plantlets can be removed as soon as they have an independent root system. Coring plants – scooping out the meristem, like removing the eyes on a potato – often results in proliferation from the basal leaves, but it is an ugly method. In another rather brutal method, which I mention only as an experiment, plants are subjected to a few hours just below freezing; this may kill them, or it may result in enormously increased offsetting.
Hybridization is not much touched on in this book, which focuses on species as they occur in nature. Here I will only mention that hybrids have a great potential for horticultural beauty and taxonomic confusion, possibly for some elucidation as well. Hybridization includes, of course, pollinating plants from different populations. When making hybrids, keep meticulous records (noting which species is the pollen parent, which is the unorthodox father), use the most attractive parents, pollinate for three or four days in a row, and don’t give up if only a few seeds form.
To date the most attractive hybrids (attractive to me, anyway) have involved H. emelyae, H. magnifica and H. bayeri, but thousands of possible combinations have not been tried, especially when one considers those involving three or more already hybridized parents! Only a few of the hybrids in cultivation have great horticultural merit; the rest should probably be eaten or otherwise recycled.
Allied to hybridization in its artificiality, but more palatable to many growers, is the practice of selection. The plants are so variable in nature that they offer limitless scope. One can pollinate the most attractive examples of a species, the seedlings can be selected and back-crossed with the parents, and in this way many (any?) desired characteristics of form or colour can be reinforced. Of course, it can not be assumed that seedlings cultivated from material from a specific locality will represent those which would have survived selection processes in their natural state. Nor is it clear how long-lived haworthias can be, either in nature or cultivation, though well-favoured plants can probably live for centuries. The little amount of this work that I have done to date is most promising. It is simply a way of beating the natural odds.
Whether or not these “artificial” beauties should still bear the data their grandparents came with is a good question; my own opinion is that they have crossed over from nature into art. Fortunately a good collection of Haworthia can represent both realms!
The Genus and Species Concept
1982 – Since Berger’s monograph in Engler’s Das Pflanzenreich, the only real contributor to an understanding of the genus as a whole, and its relation to the Aloineae has been A.J.A. Uitewaal. This is expressed in 2 series of papers published in Desert Plant Life (1947), Cactus and Succulent Journal of Great Britain (1947), Succulenta (1948) and Sukkulentenkunde (1951). Originally Haworth recognised only Linnaeus’ genus Aloe and it was Duval (1809) who erected Haworthia to contain the species in Haworth’s section Parviflorae. According to Uitewaal, Haworth and Willdenow independently applied the same name Apicra to the genera now recognised as Astroloba and Haworthia respectively. Apicra Willd. is thus a synonym of Haworthia Duval, and the name Apicra Haw. for a different genus is a later homonym and invalid. This is the reason for the creation of the name Astroloba Uitewaal. Actually Haworthia is already a nomen conservandum in favour over the earliest name Catevala used by Medicus (1786).
Apart from Salm-Dyck, no authority with any reasonable knowledge of the Aloineae has suggested that the genera do not fairly reflect the diversity of the group. Parr has published (African Succulent Plant Society, 1971-2) what purports to be a revision of the genus Astroloba in which he includes these species together with Poellnitzia Uitew. as the section Quinquefariae in Haworthia. The wisdom and intent of such a change is dealt with by Bayer (National Cactus and Succulent Journal, 1972) and it is rejected for a number of reasons including that re-alignment of genera when the elements within them remain poorly defined, is unquestionably premature. Existing genera do portray natural groups despite weak rationalisation in terms of morphological and other criteria. The independent recognition of the different status of Haworthia and Astroloba by Haworth and Willdenow is indicative of the ‘naturalness’ of the two genera. It is true that there was some confusion as to the relation of H. viscosa (L.) Haw. to Astroloba prior to Haworth. Mrs P. Roberts-Reinecke in an unpublished revision of Astroloba (M.Sc. thesis, Department of Botany, U.C.T.) also concludes fairly convincingly that Apicra aspera Haw. is in fact synonymous with H. nigra Haw. This is nevertheless easily attributed to poor observation of floral characters and doubtful evaluation of vegetative characters. The question of incorporation of Astroloba in Haworthia needs consideration in light of recognition of three subgenera in Haworthia. This first finds expression under Salm-Dyck, later Berger, then Uitewaal (1947) and then Bayer (1971). The distinction between Haworthia and Astroloba on the basis of bilabiate as opposed to stellate flowers is extremely weak and Uitewaal’s usage of ‘approximately’ (plus/minus) in his key is very apt.
Mrs. A.A. Obermeyer-Mauve in Bothalia (11:119, 1973) included Chortolirion Berger (one species only) in Haworthia, and Chamaealoe Berger in Aloe L. She at the same time endorses Parr’s inclusion of Astroloba and Poellnitzia in Haworthia. The sole reason given here is also that the latter genera have regular flowers while some haworthias do too, and the claim is made that there are no correlated distinctions. This is quite a fallacious argument for reasons expressed elsewhere (Bayer, 1971, 1974). Mrs Mauve likens Chortolirion to H. graminifolia Smith when in fact the latter is not the least bulbous and neither is there the degree of similarity in the leaves which she suggests. The flower of Chortolirion is like that of the Haworthia subgenus Hexangulares and cannot be confused with that of the subgenus Haworthia where H. graminifolia belongs. In The Genera of Southern African Flowering Plants by R.A. Dyer (1976), Astroloba, Poellnitzia and Chortolirion are all included in Haworthia. For the purpose of this book the traditional generic states are maintained.
The species concept in Haworthia is discussed briefly by both Uitewaal and Resende who stress the view that speciation in the genus is still in progress, and that species are poorly defined. To a greater or lesser degree this must be true in the majority of plant genera, and is certainly not true for all species, even in Haworthia. Each species needs to be considered in terms of distribution, variability and isolation. Speciation and the evolution of discrete segregates (species, subspecies, varieties, etc.) is self-evidently a function of isolationary mechanisms. Whether anyone can honestly claim to have an adequate proven explanation for sympatric distribution of so many Haworthia species in so many areas, each with specific habitat requirements and consequent discontinuities is very doubtful. The isolationary mechanisms operative where species are growing in the same near vicinity can be of several kinds. Either the plants are not cross-fertile due to physiological or genetic barriers; or the fertile seed does not produce a seedling which can survive ecological selection pressure; or flowering times are staggered. Experience suggests that hybridisation is a common phenomenon, at least in cultivation, and many ‘species’ are, by admission, garden hybrids. Although hybridisation in the field is far less common than this suggests, it does occur and it can be implied that species have arisen from such hybrids too. Selection as a result of ecological requirements is in the writer’s opinion, a highly acceptable explanation for the development of Haworthia species whether breeding barriers occur or not. Flowering time also differs consistently enough in the field to suggest that it is a good isolating mechanism. However, the species problem in Haworthia very seldom arises where species grow together, but rather from the difficulty in associating populations strung out in isolated localities. These populations can be mostly recognised as species complexes, but populations do occur which cannot be confidently assigned to either one or more of their geographically obvious counterparts. The complexity of what species are is beyond the object of this book, beyond providing some clue to the concept of a species accepted in the checklist. The species have been largely defined in terms of geographical distribution, on the assumption that selection across ecological gradients is mainly responsible for their origin. It is very comforting for the laymen to know that even the most learned botanists cannot define a species in simple terms. It is important to realise, though, that a species is more than a simple expression of differences between plants which the ordinary man in the street can recognise. Two plants may appear to be quite different in size, colour, markings and flower, yet easily recognised as the same kind (species) when seen growing together with a number of other plants sharing all the differences of the first two. If one or several plants still stand out from such a group, then they can be recognised as distinctive forms. Normally a plant species can and should be considered to consist of several groups (populations) growing at different sites (localities). If one of these groups is considered to be sufficiently different from the others, then it may earn recognition as a variety. If several groups are different in this way, then they may be referred to as subspecies. In each case the degree of difference is important and different people may not share a decision to recognise differences by naming forms, varieties, subspecies or even species. In this way classification and taxonomy can become distorted by the judgement of the individual applying himself subjectively to the naming and renaming of plants. The more information available, and the more thoroughly and objectively the work is done, the less likely is the final product to be a reflection of individual idiosyncrasy and personal opinion. The differences between plants of the same kind may result from external factors such as soil type, exposure to light, availability of water, etc., or from internal factors such as age and genetic make-up. Uitewaal expressed the view that it would be possible to classify Haworthia even without a full understanding of how these factors actually affect the plants. The author agrees with this view because experience shows that under moderate conditions of cultivation, plants can be expected to remain consistent. Uitewaal in 1951 was perhaps less optimistic that floral characters would be useful than he had suggested in 1947, but the importance of using flowers as a source of characters is not diminished thereby. Most authorities have claimed that floral characters are too uniformly the same to be of any use in classification. Such a conclusion may have arisen only because similarities occurred where differences were expected and vice versa, based on the a priori presumption that classification by vegetative characters was correct. Vegetative characters have always been the predominant basis for classification in Haworthia in complete contrast to other genera. Far too much weight has been attached to differences which may occur even between plants of the same kind at the same locality. Examples are the presence or absence of tubercles or spines, or the number of veins showing on semi-transparent leaf faces. Smith’s descriptions are models of detail and uniformity, but rather than circumscription of species and varieties, are descriptions of single plants. Many other descriptions include few diagnostic characters by which the identity of a plant among near relatives can be determined with any certainty. The descriptions are padded with measurements which have no reference to the range of dimensions in the entity, or by characters equally applicable to other, or all, members of the genus. The state of Haworthia classification is easily assessed by the past arrangement of the species in 20 sections compared with their natural disposition in the three subgenera. If even sections have not been defined correctly to include related species let alone variants of one species, there can be little confidence that the species themselves are all correct.
1999 – As far as genera are concerned, the attempt to relegate Astroloba, Poellnitzia and Chortolirion to Haworthia appears to have failed, so that good sense can be said to be prevailing. Dr G.F. Smith has written many papers relating to Aloaceae which generally project the view that the genera are adequately different. Emphasis on the differences within Haworthia at a corresponding level is still sadly lacking and Dr L.E. Codd’s comment that ‘small differences are going to be ignored’ is sadder still. Small in size does not equate to small in significance. There can be no doubt that Haworthia is composed of three very distinctive sub-groups, recognisable on floral characters alone. I have previously commented on the similarity of the Chortolirion flower to that of the Hexangulares. I would like to say forcefully that while I doubt if I could distinguish many of the species in the subgenus Haworthia on the basis of the floret, I would confidently attempt to recognise Chortolirion among them, and among any of the other subgenera too. Nevertheless a taxonomic revision is only an hypothesis which cannot be proved right. If tested and it is infirm, a new hypothesis should replace it. Thus there will be an on-going attempt to re-define the genera, and one can only hope for some balanced approach where due recognition is given to the foundations of the old, before an equally, or even more greatly flawed alternative is put forward. The subdivision into lower groups (Sections) is very difficult and if the prime divisor is disregarded (i.e. the flower), likely to be impossible. If the subgeneric characters are disregarded, on what kind of characters are the species to be recognised? Simple characters which one would expect to separate species, and form sectional characters, do not exist. For example tuberculate versus smooth leaf surfaces or leaves spineless versus spined, frequently occur within species. Even windowing or coloration of the leaves is not a certain clue to identification. Ultimately the test is distribution and co-occurrence.
Why the taxonomy of these genera in the Aloaceae has been considered more confused than elsewhere in the plant kingdom is a bit of a mystery to me. I see a similar situation in the Cactaceae (although I do not know it very well); and have experienced the same problems in Asparagus, Oxalis, Pelargonium, several other genera which I came to know quite well, the Stapelieae, and also strangely enough in Noctuid moths. Very high collector interest and low academic involvement may be seen at the root of the problem. I am not confident that intellect as such will necessarily produce more truthful answers. The concept of truthfulness I use here is not that of an honesty of expression, but that of objective factual correctness. Anything subjected to the intense interest of collectors, is bound to generate controversy. It is salutory to see what wars have been waged in very quiet backwaters of botanical interest, between protagonists barely known to any but each other. I would like to repeat what I have already said in the introduction where I credit collectors with as much acumen as any botanist who claims that title by virtue of qualification rather than from interest. Both groups rate from low to high on the various and many scales by which intellect could or should be measured, and with an unfortunate poor correlation to how the individuals may assess themselves.
To understand Haworthia, it seems to me that James Gleick’s book on ‘Chaos: Making a New Science‘ is useful reading. Even if intellectually incomprehensible, the gist of infinite variability, period doubling and bifurcation diagrams will help how one thinks about species. The nature of variation in plant species – the nature of genetic material, the way it is ordered, and the way in which this is expressed in the appearance of plants – can be said to be a phenomenon of chaos theory: disorder which is highly ordered. We look for regularity and order in plant species and populations and it simply is not visibly there. How many of us have said that our classification system does not match the facts?
Vavilov’s law of homology presents the view that variation in one element of a group is parallelled in other elements of that group. He said that: ‘In closely related species and genera, similar series of heritable variations occur. These are found so regularly that knowing the series of forms within the limits of one species, we can predict the occurrence of parallel forms in other species and genera’.
Our attempt to assign position to things is really to locate them on a kind of dendrogram. No two individuals in a sexually propagating population are exactly the same, and the same can be said of vegetatively propagated plants, with certain provisos. Thus at what time will we recognise that a species has split into two? We have to do it on some kind of statistical basis or make an ‘educated’ guess. A split may be the consequence of a difference of only a tiny fraction of the total genetic material. A dendrogram indicates the split, but the appearance of the individuals in the now-divided species may not. On the other hand a single gene character may induce a massive change in appearance which might occur frequently or infrequently within a population. So it is logical and reasonable that individuals of closely related species will tend to resemble one another, and that variation within a species may exceed that between species. Some individuals of different species may resemble each other more closely than they resemble individuals within their own ranks.
This book has of necessity set out to honestly bypass technical nicety and complexity and to practically identify nodes in a complex interlinked web. I do not believe that there is any way that the vast variation that does exist can possibly be accounted for in any written or pictorial record. In many instances there are no real boundaries and we have adopted the view:
that species are a group or groups of interbreeding or potentially interbreeding individuals varying continuously, genetically and morphologically, in both time and space.
There is an island-like situation in Haworthia which is both real in terms of the isolation of habitats, and also artificial due to destroyed habitats. The latter are significant to us only in present time because they affect what we can observe. The observation and interpretation (measurement and evaluation) of continuity is limited by inaccessibility to the past and by the sheer physical problem involved in locating and seeing everything. It is the nature of creation that diversity exists in space and change is brought about in time. Chaos theory must apply.
In this new treatment I have tried not to get involved in the rigours of systematic nomenclature for which I am not adequately proficient nor sufficiently orthodox. It has been proved to me that it is subject to gross error and personal antagonism as any other field of human endeavour. Some people are able to indulge in a semantic and legalistic forest of words for that reason and pleasure alone. I respect the purpose, but it is worth recounting briefly how senselessly it has been applied to Haworthia. In my very first encounter with the problem of the name H. margaritifera vs H. pumila I put the problem to a Professor and two of his doctoral graduates. None would even brave an opinion. Then Dr Codd offered a view applied by Col. Scott, which was up-ended by Dr Wijnands. Since then that has been brought into question too, and I have become convinced that argument is directed to the end-point desired. Rationalization follows decision. In truth it appears they have all missed the mark because the element they are most frequently arguing about is most probably what we commonly refer to as minima and not pumila. Thus the correct interpretation is perhaps that pumila should be scrapped and that maxima should be used for this larger species, and margaritifera for the littler minima. An attempt in this direction will be apparent in this work, with an apology to the cognoscenti who are most active in critical retrospect, rather than pro-actively helpful when needed.
We have the case of the argument against the retention of emelyae based on doubts as to origin of the type, despite evidence which exists to show the contrary. I was no doubt wrong in avoiding the use of atrovirens for H. magnifica var. maraisii, and am also argued to be wrong about integra. In my view these are cosmetic problems which should not be allowed to distract anyone until such time as someone can come along with a truly better understanding of the things they are trying to classify. There seems little sense in getting all the names right while the elements to which they refer remain totally confused. There is a real problem in trying to typify the species on the basis of old herbarium specimens and also on the basis of old illustrations, because quite simply it is often very difficult to even identify living flowering material without direct reference to origin. In this respect it should be noted that the whole question of typification has been addressed by I. Breuer and D. Metzing and shown to me in manuscript form as I finished this manuscript. I assume that their typification is authoritative and where I have depended and used their typification it is acknowleged by the letters “B&M” as the source for designation of the type. I do not consider their typicication to be based on any real knowledge of the genus, and their selection of neotypes and epitypes is often unfortunate. This is apart from the lecotypification of photographs in the Berlin Herbarium which were mostly published with the original descriptions. Otherwise I have considered the types to be as designated in this work, and representing the historical record and the traditional interpretation and understanding of the names.
It has not been necessary for me to have the view confirmed that species of Haworthia can only be reasonably defined in terms of geographical distribution and interpreted on the grounds of co-occurrence. The nature of the interlinking web has been enormously strengthened by the collections I have seen in the past 15 years, without seriously weakening the artificial structure of names. It is this ‘structure’ which I would claim has facilitated communication and observation. The discussion under the individual species will deal with new collections and the insights which they bring.
The main point is that there are alternative decisions that can be made or could have been made – and if good sense is maintained there will be no problem. Problems arise from unseen mistakes as we have in the confusion of H. arachnoidea sensu Scott which is the same species as H. herbacea sensu Bayer. This is not just the product of nomenclatural nicety. There is also the mistaken identity of H. serrata by Col Scott where he has assumed that a population of H. mucronata is that species. In this revised edition, an attempt will be made to specify a type for each name, following Breuer and Metzing as closely as possible.
Other difficulties arise from completely different perceptions about what constitute species. Unless a good strong base is laid in the geographic component, it just becomes impossible to handle the close resemblances within Haworthia and the ‘chaotic’ nature of transitional populations. In this treatment we find difficulty in supporting, say, H. reddii as a species because we do not regard it as substantial enough to hold that rank. We think a species should be more substantial and have a range of distribution and variation across that range. We are, I think, a little pretentious in the use of the many ranks of subspecies, variety and form. In this revision I have reverted to mostly the use of varietal rank. However, this does have its own problems because as soon as a subordinate rank is created, it implies that the species is composed of two discrete elements. This is probably not problematic where subspecies are concerned and the concept of extended variability is retained. However, when a variety is selected to illustrate or communicate a specific population which is unusual in one or other respect the balance of variability is loaded towards the typical and we have incongruity. This is the view adopted here i.e. there is not a typical variety in the sense demanded by the nomenclatural system. Rather there are variable species which intergrade, and among them are consistencies which are identified as distinctive varieties. The convention of citing locality is by far the most suitable way of adequately communicating where identification becomes doubtful, as it inevitably will. The ideal of consistency is very difficult to attain because the plant populations seem to be aligned at different grades of similarity.
I recognise that if my initial view of the species is reasonable, then the formal recognition of varieties may ease the understanding of the nature of the variability and existing or potential inter-relationships between the species. This will be particularly evident in the way variation of the old H. retusa varieties are now treated and also in the many varieties recognised here, particularly in H. archeri and H. heidelbergensis, which were relatively unknown in 1983. Similarly while several new species appear in this work as a product of new finds, new insights, corrected mistakes, and new mistakes, some old species have been relegated to varietal level for the same reasons. Lesser ranks should not be taken too seriously as they also represent a view which changes with familiarity and with time. A revision may present a new set of names and ranks, but does not erase the history of those names and they remain there for the edification of all.
Synopsis of Taxonomic Changes
The main changes in this revised edition are as follows:-
- H. aranea A.Berger = H. arachnoidea var. aranea (A.Berger) M.B.Bayer.
- H. archeri Barker ex M.B.Bayer = H. maru.miana var. archeri (WF.Barker ex M.B.Bayer) M.B.Bayer.
- H. batesiana Uitewaal = H. marumiana var. batesiana (Uitewaal) M.B.Bayer.
- H. comptoniana G.G.Sm. = H. emelyae var. comptoniana (G.G.Sm.) J.D.Venter &: S.A.Hammer.
- H. correcta of Scott (pro parte H. emelyae V.Poelln.) = H. bayeri J.D.Venter & S.A.Hammer.
- H. divergens M.B.Bayer = H. monticola Fourc.
- H. graminifolia G.G.Sm. = H. blackburniae var. graminifolia (G.G.Sm.) M.B.Bayer.
- H. habdomadis V.Poelln. var. habdomadis = H. mucronata Haw. var.habdomadis (V.Poelln.) M.B.Bayer.
- H. habdomadis V.Poelln. var. inconfluens (V.Poelln.) M.B.Bayer = H. mucronata Haw. var. inconfluens (V.Poelln.) M.B.Bayer.
- H. habdomadis V.Poelln. var. morrisiae (VPoelln.) M.B.Bayer = H. mucronata Haw. var. morrisiae (V.Poelln.) M.B.Bayer.
- H. maughanii V.Poelln. = H. truncata var. maughanii (V.Poelln.) Fearn.
- H. magnifica var. major (G.G.Sm.) M.B.Bayer = H. emelyae var. major (G.G.Sm.) M.B.Bayer.
- H. magnifica var. paradoxa (VPoelln.) M.B.Bayer = H. mirabilis var. paradoxa (V.Poelln.) M.B.Bayer.
- H. poellnitziana Uitewaal = H. minima var. poellnitziana (Uitewaal) M.B.Bayer.
- H. pumila (L.) Duval sensu C.L.Scott = H.pumia (L.) M.B.Bayer.
- H. radula (Jacq.) Haw. = H. attenuata var. radula (Jacq.) M.B.Bayer.
- H. retusa var. acuminata M.B.Bayer = H. magnifica var.acuminata (M.B.Bayer) M.B.Bayer.
- H. retusa var. dekenahii (G.G.Sm.) M.B.Bayer = H. magnifica var. dekenahii (G.G.Sm.) M.B.Bayer – pro parte.
- H. retusa var. argenteo-maculosa (G.G.Sm.) M.B.Bayer = H. pygmaea var. argenteo-maculosa (G.G.Sm.) M.B.Bayer – pro parte.
- H. rycroftiana M.B.Bayer = H. mucronata var. rycroftiana M.B.Bayer.
- H. starkiana VPoelln. = H. scabra var. starkiana (VPoelln.) M.B.Bayer.
- H. translucens Haw. sensu M.B.Bayer = H. gracilis V.Poelln.
- H. unicolor (V.Poelln) unicolor = H. mucronata Haw. var. mucronata.
- H. unicolor var. helmiae (V.Poelln.) M.B.Bayer = H. mucronata var. mucronata Haw.
- H. unicolor var. venteri (VPoelln .) M.B.Bayer = H. arachnoidea var. nigricans (Haw.) M.B.Bayer.
- H. wooI!eyi V.Poelln. = H. venosa subsp. woolleyi (VPoelln.) M.B.Bayer.
- H. xiphiophylla Baker = H. arachnoidea var.xiphiophylla (Baker) M.B.Bayer.
New species included are:
- H. aristata Haw.
- H. bayeri J.D.Venter & S.A.Hammer.
- H. maraisii V.Poelln.
- H. outeniquensis M.B.Bayer.
- H. pungens M.B.Bayer.
- H. vlokii M.B.Bayer.
Species desclibed since publication of The New Haworthia Handbook and their treatment :-
- H. joeyae CL.Scott = H. cooperi Baker var. dielsiana (V.Poelln) M.B.Bayer.
- H. magnifica VPoelln. var. splendens J.D.Venter & S.A.Hammer.
- H. mcmurtryi C.L.Scott = H. koelmaniorum C.L.Scott var. mcmurtryi (C.L.Scott) M.B.Bayer.
- H. pehlemanniae C.L.Scott = H. nortieri var. pehlemanniae (C.L.Scott) M.B.Bayer.
- H. pringlei C.L.Scott = H. decipiens V.Poelln. var. pringlei (C.L.Scott) M.B.Bayer.
- H. reddii C.L.Scott = H. cymbiformis (Haw.) Duval var. reddii (CL.Scott) M.B.Bayer.
- H. venusta C.L.Scott = H. cooperi var. venusta (C.L.Scott) M.B.Bayer.
In my view they are not substantial elements with variation and range, and can be accommodated within species already extant. The illustrations in this work will include similar elements in other species and still fall short of accommodating quite substantial variation in many of the species.
Key to the Subgenera
1982 – Uitewaal (1947) presented a Latin diagnosis for two sections, Triangulares (type species Haworthia translucens Haw.) and Hexangulares (type species H.coarctata Haw.). The latter section he again divided into the Robustipedunculatae and Gracilipedunculatae, partly derived from Berger (1908). Uitewaal did not relate this subdivision to then existing sections and stated specifically that it was only of a tentative nature. The following exposition (Bayer, 1971) takes Uitewaal’s division to its logical conclusion with the recognition of three subgenera. In the process, the subgenus containing the type species for the genus repeats the name of the genus:
1. Perianth at base triangular or rounded-triangular, style upcurved, tube obclavate ………. subgenus Haworthia
Type species H. arachnoidea (L.) Duval
– Perinath at base hexangular or rounded-hexangular, style straight, tube obcapitate ………………….
2. Perianth gradually narrowing to junction with pedicel (substipitate), tube curved…….. subgenus Hexangulares Uitewaal ex M.B.Bayer
Type species H. coarctata Haw.
3. Perianth abruptly joined to pedicel (non-stipitate), tube straight ………………. subgenus Robustipedunculares Uitewaal ex M.B.Bayer
Type species H. pumila (L.) Duval
This construction of subgenera would necessitate re-organisation of the section Margaritiferae as composed by Jacobsen (1965), as several of those species belong naturally with the Coarctatae. H. papillosa (Salm Dyck) Haw. was placed in the section Coarctatae by Baker on account of its elongated stem. This was totally incorrect as caulescence does occur in the Margaritiferae where the plant can obtain sufficient water and nutrient to maintain an extended growing point. More often the central parent stem dies away and the plant is perpetuated by several acaulescent offsets. However, there are many other anomalies which can be quoted in demonstrating the inaccuracy of the sectional divisions up to Jacobsen (1965). The real problems and difficulties in Haworthia are best appreciated by referring to synonymies of species like H. reticulata and H. herbacea. Here it is seen how authors have described several species from what can be proven to be a single entity.
1999 – A key to species seems to be the summum bonum of revisionary work and it would be really an achievement to provide such a key by which the species could be readily identified. In Haworthia I do not think a key will provide a probability for correct identification of existing material much better than 50% of the time. For new collections from poorly explored or poorly collected areas, it may even be worse. This may confirm that in reality there are many fewer species, which I would not dispute.
The most common and practical way of identification is by trial and error using visual comparisons. In my experience there have been and are misidentifications with the simplest material and even the best of keys and explanatory notes. There are enough cases where even photographs and illustrations are hopelessly misidentified.
There have been several attempts to create sections in Haworthia and these have nearly all been across the subgeneric boundaries. One might expect that sub-division would be easier if the subgeneric limits were first accepted, but the subgenus Haworthia seems to defy resolution. Whatever way is attempted is eventually confounded by affinities across demarcated boundaries. Some links and affinities are clearly stronger than others. It is clear to me that as exploration and collecting yield more material, so will the complexity of the problem. Hence the hope of finding a simple solution will recede with time and not as we like to think, improve.
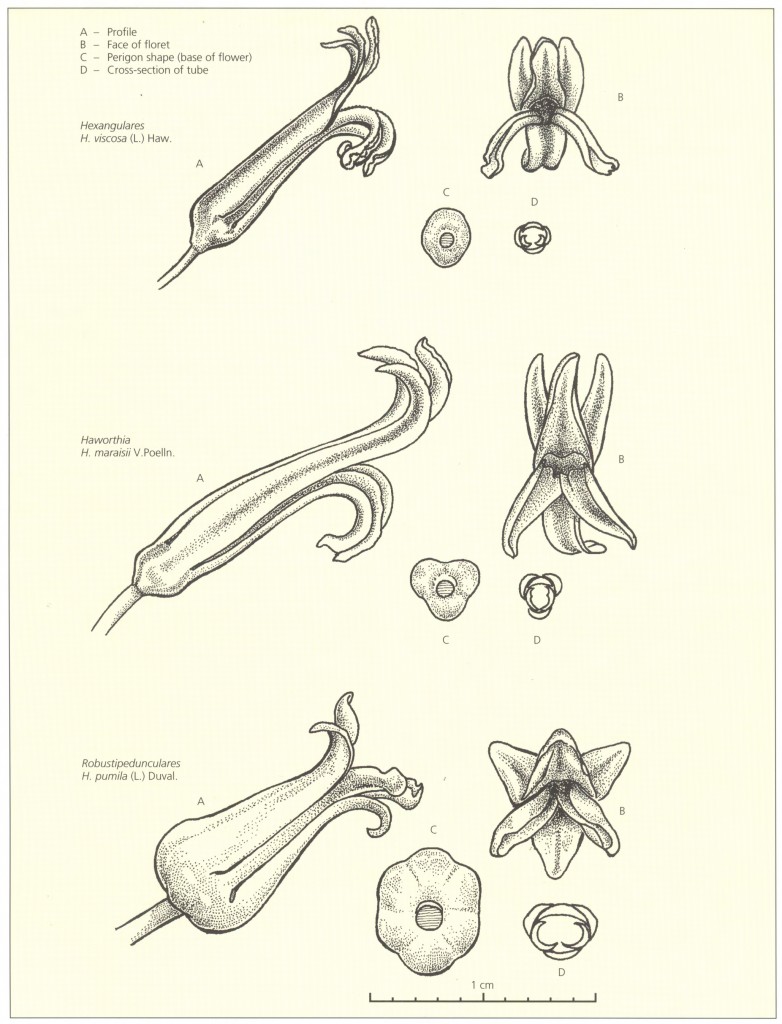

Pingback: Mystery - part 30, mirabiloids - Haworthia Updates
Pingback: 陽台上的住客001—鷹爪草屬搞分裂? - 分類沙丘