2. Haworthia arachnoidea (L.) Duval, Pl.Succ.Hort.Alenc. :7(1809). Haw., Syn.Pl.Succ. :96(1812). nec Pole Evans, Flow.Pl.S.Afr. 7:t.248(1927). Bayer :97(1976). Bayer :27(1982). nec Scott, Cact.Succ.J(U.S.) 49:205(1977). nec Scott, Aloe l6:41(1978). nec Scott :66(1985): Aloe pumila var. arachnoidea L., Sp.Pl. 1:322(1753): Aloe arachnoides Thunb., Diss. :311(1785). D.C., Pl.Gr. :f.50(1799). Ker-G., Bot.Mag. :t756(1802). Haw., Trans.Linn.Soc. 7:10(1804). S.D., Monogr. 12:t.2(1840). Lectotype: icones :78,t27, Comm., Prael.Bot. :78(1703). Epitype (B&M): CAPE-3319 (Worcester) Buitenstekloof (-DB), Bayer 153 (NBG). H. arachnoidea var. minor Haw., Suppl.Pl.Succ. :52(1819). Type: Not preserved.
arachnoidea: spider-like.
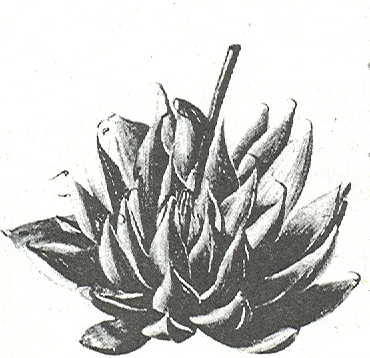
Rosette stemless, variable in size from 60 to exceptionally 120 mm φ, solitary or forming small clusters. Leaves 25-80 dense incurving, uniformly light to dark-green, no translucence and only occasionally faintly reticulate, flattened and often drying grey‑white to brownish at the tips forming a protective cover for the leaves. Triangular‑ to ovate‑lanceolate, 20‑70 X 10‑15mm, keeled, margin and keels bearing translucent spines to 12 mm in length, apex acuminate‑aristate. Inflorescence to 300mm. Flowers 20-30, perianth white.
1982 – There is some controversy and confusion over the application of the name H. arachnoidea. The difficulty stems from the interpretation of an illustration dating back to 1703. However, the interpretation arrived at by Salm-Dyck appears reasonable and in the light of his illustration it is difficult to understand why H. setata was not earlier reconciled with this species. H. arachnoidea is recognised by its uniformly plain green leaves. The ‘cobweb’ suggested by the name, is derived from the many white hairs on the leaf margins and keel, which in a healthy well-grown specimen do indeed resemble a spider-web. However, Ker-Gawler (1804) did not like this name ‘cobweb aloe’ given by Miller (1768) because he considered that the spined leaves more resembled the legs of a spider and hence the name. The plants are variable in size even within populations. Normally they do not exceed 60 mm in diameter but in some populations magnificent specimens of over 120 mm diameter may occur. The distribution does not coincide distinctly with any of its likely relatives and a great deal more information will be required before its relationship to any other species is better known. H. arachnoidea occurs in Namaqualand and in the Ceres Karoo. It is replaced on the Roggeveld Plateau near Sutherland and then eastwards, by H. semiviva. Plants from the Merweville area suggest an affinity with H. decipiens but are without the rather broader, flatter leaves and more deltoid spines characteristic of that species. H. arachnoidea occurs recognisably with H. archeri at Matjesfontein. Forms further to the southwest have marked leaves and are a deeper purplish-green so that there may be some difficulty in reconciling them with H. arachnoidea. There is no difficulty in recognising H. arachnoidea in the Worcester/Robertson Karoo, where it is quite common. It is poorly known in the Montagu area apart from at Cogmanskloof, Baden and the Ouberg Pass. A rather depauperate form is found in the Tradouw Pass south of Barrydale, and in the wider Ladismith area it is replaced by either H. unicolor or by H. habdomadis. However, near Ladismith itself H. arachnoidea is small and densely white-spined – more coarsely so than in the Oudtshoorn area where the plants are often small and the spination is finer and a little softer too (see H. aranea). A weaker spined but very proliferous form occurs in Meiringspoort, east of Oudtshoorn. The distribution eastwards from here is not clear. H. decipiens occurs at Klaarstroom north of the Swartberg mountains and it is possible that odd collections east of Uniondale, attributed to H. arachnoidea may in fact be either this species or H. unicolor. The illustration in Flowering Plants of South Africa (1927) is clearly of H. cooperi. H. arachnoidea is generally not proliferous and offsets very slowly if at all. Its distribution is clearly that of a winter-growing species and its occurrence on south slopes and shaded under bushes suggests the treatment it should get in cultivation.
1999 – The 1703 illustration referred to above is the illustration t27 in Commelin’s Praeludia Botanica. Bayer (in Excelsa 12:91, 1986) discusses the problem of interpretation surrounding this illustration and the associated confusion, pointing out that Scott’s analysis of the picture does not support his argument. Thus Scott uses this illustration to typify his concept of H. arachnoidea which is the same as Bayer’s H. herbacea in this work. Kerr-Gawler disliked Miller’s name ‘cobweb’, preferring the likeness to the spined leg of a spider rather than to the woolliness of its web. General usage seems to have favoured the use of ‘arachnoidea’ in terms of woolliness rather than simply spiny. If one looks at the illustration Botanical Magazine t.1417 that is used by Scott to typify his concept of H. translucens (viz. Aloe arachnoidea var. translucens (Haw.) Ker-G.) the situation is further confounded. The illustration in black and white indicates a species vegetatively fairly similar to the t27 of Commelin. Scott applies the epithet to what, in terms of its reported location, can only be one of three species viz. H. arachnoidea (H. setata sensu Scott), H. pehlemanniae or H. marumaina var. archeri (H. marumiana sensu Scott). The herbarium specimen referred to by him is certainly of the latter, while the illustration of his H. translucens appears to be of H. arachnoidea. It is not clear what Haworth actually intended in his Revisions when he created the section Arachnoideae and omitted mention of H. arachnoidea. I assume that he simply subsumed it under his H. setata. When Salm-Dyck discussed Aloe setosa he seemed to suggest that it was a remnant not adequately covered by Haworth’s description. Typification and usage of these old names is problematic and the scheme followed here does not have a flawless rationale. However, Salm-Dyck’s illustrations of both Aloe arachnoides and Aloe setosa are certainly not of H. herbacea as perceived in this work. In ascribing the type to the Worcester/Robertson Karoo there is an inherent problem in that elsewhere there will be a residue which may not be well accommodated in other varieties. However, the name setata is taken up for the more centrally situated variety which will better do that.
The varieties are largely geographic entities. The discovery of H. nortieri var. pehlemanniae (Scott) Bayer, as well as that of variants of H. marumiana var. dimorpha, which are vegetatively practically inseparable from H. arachnoidea, create problems. Var. pehlemanniae grows in very close proximity to H. arachnoidea var. scabrispina from which it quite distinctive, but it is very difficult to distinguish it from some of the other varieties without flower or habitat information. Possibly the former has leaves which tend to be more flaccid and thicker when fully turgid. We already know that H. mucronata var. mucronata may probably be the origin of arachnoidea-like plants in the Tradouw Pass. A similar transition of a deviant population of H. mucronata from north of Montagu, to the Cogman’s Kloof, also occurs. In the widest context, H. arachnoidea is impinged on by all three varieties of H. nortieri, as well as by H. marumiana var. archeri, H. mucronata, and H. decipiens. It is quite probable that correctly interpreted, more than one species may be involved in the concept of H. arachnoidea given here. However, there is no reason why over its whole range it should not exhibit the same degree of variation that for example H. magnifica or H. turgida exhibit over their comparatively restricted ranges. The species does hybridise with H. blackburniae var. blackburniae and var graminifolia, as well as with H. truncata.
a. var. arachnoidea
The form common in the Worcester/Robertson Karoo is taken as the typical variety although the choice is arbitrary. In this area there is no confusion with other species and although it grows in close association with a number of other species, there is no evidence of hybridisation. This variety often occurs on southern slopes where it is embedded in moss and lichen, and thus very moist in the winter months. The plants can reach as much as 18cm in diameter. The basic leaf coloration is darkish-green and the marginal spines are white. The terminal bristles tend to be blackish.
Distribution: 3319 Worcester): Hex River (-BD), Bruyns 2411 (NBG); Tonnel Stn. (-BD), Bayer in KG36/71 (NBG); Worcester (-CB), Fouche 14 (PRE); 8km NE. Worcester (-CB), Bayer in KG 640/69 (NBG); Worcester (-CB), Marloth 12572 (PRE), Venter 3 (BOL); Effata (-DA), Bayer in KG 118/70 (NBG); Buitenstekloof (-DB), Bayer 153 (NBG).
-

Haworthia arachnoidea var. arachnoidea JDV86/109 east of Worcester. The typical variety occurs in the Worcester/Robertson Karoo and the end-awn tends to be blackish.
b. var. aranea (Berger) Bayer comb.nov.
H. bolusii var. aranea Berger, Das Pflanzen. 33:114(1908). Scott, :72(1985), not as illustrated. H. aranea (Berger) Bayer :98(1976). Bayer :29(1982). Lectotype: icon :114,t39, Berger, Das Pflanzen. 33(1908). Epitype (B&M): CAPE-3322 (Oudtshoorn): Moeras River (-CA), Bolus 12372 (BOL).
aranea: cobweb.
1982 – This species is described and illustrated in Berger’s revision. The first comparable plants seen by the writer were from the fynbos vegetation between Oudtshoorn and Uniondale. The uniform green coloration and soft canopy of hairs, as well as the existence of larger similar forms near Oudtshoorn itself, suggested an affinity with H. arachnoidea rather than with H. bolusii. Since then a much smaller proliferous form of H. bolusii has been discovered by Peter Wilkins east of Jansenville which could well be closely related to H. aranea. Here the plants have the typical bluish green colour and translucence of the upper leaves as in H. bolusii. They occur in a flaking blue shale. Plants close by in compacted brown shale are larger and with fewer firmer hairs. It appears more likely that Berger’s plants originated from the Oudtshoorn area. Until the complete picture of species distribution is available H. aranea is being retained.
1999 – No new information suggests any change here and it has long been known that it intergrades with H. arachnoidea var. setata around Dysseldorp and De Rust as well as in Schoemanspoort. Thus H. arachnoidea in itself is so variable that the pressure for consistency seems to call for varietal rank. Col Scott maintains this element as a variety of H. bolusii and cites a specimen which is apparently not that species at all, disregarding the comments made above. Scott’s illustration also does not accord with the type of var. aranea species but nevertheless appears to represent H. arachnoidea. The earlier comment ‘until the complete picture…’ is nonsense and it is unlikely that there can be new evidence to prove the original species closest to either bolusii or arachnoidea; nor likely or even possible that distribution data will clarify a position clouded by history. This variety is characterised by its small size (to 6cm diam.) and very dense soft spination. Despite the comments made in 1982 concerning colour and translucence, there opacity of the leaves of this variety (and the species) is often forgotten.
Distribution: 3321 (Ladismith): Huis River Pass (-DA), Bruyns 1266a (NBG). 3322 (Oudtshoorn): S. Cango Caves (-AC), Bayer 4462 (NBG); Raubenheimer Dam (-AD), Bayer 4647 (NBG); Jagersrivier (-BD), Rossouw 411 (NBG); Moeras River (-CA), Esterhuysen 19497 (BOL, PRE), Bolus 12372 (BOL); Moeras River (-CC), Barker 7720 (NBG); De Rust (-DA), Kleinberg Forest (-DA), Matthews 1211 (NBG); Smith 4008 (NBG); Ganskraal (-DC), Fourcade 3398 (BOL), Stayner 174 (BOL), Bayer 2086 (NBG). 3324 (Steytlerville): Springbokvlakte (-BC), Bruyns 1827 (NBG).
-

Haworthia arachnoidea var. aranea JDV90/59 Vergelegen. Always with very narrow leaves and soft spines. -

Haworthia arachnoidea var. aranea JDV97/108 north of Schoemanspoort. The accumulation of old black leaves is very characteristic.
c. var. namaquensis var.nov.
Type: CAPE-2917 (Springbok): Karrachabpoort, Richtersveld, Bayer 1674 (NBG, Holo.).
namaquensis: from Namaqualand.
Rosette as for species, to 60mm φ. Leaves paler green. (A var. arachnoidea foliis subviridibus et in terra valde submersa differt).
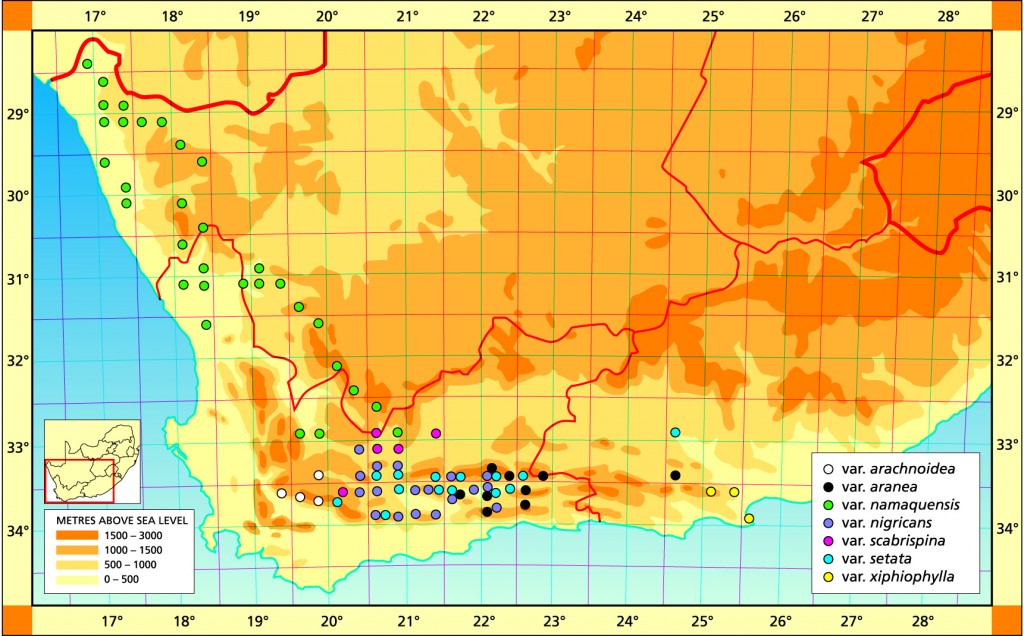
The var. namaquensis has not been previously accommodated. It is essentially recognised for its geographic association and applies to populations in Namaqualand, Bushmanland and the Ceres Karoo. The spination is usually moderate and the plants are usually lighter coloured than elsewhere in the species. It also differs from the typical variety in being pale green and well-drawn into the soil.
Distribution: 2816 (Alexander Bay): Top Hellskloof (-BD), Roux 519 (BOL). 2817 (Vioolsdrift): Cornellsberg (-CA), Bruyns 3305 (NBG); N. Lekkersing (-CC), Venter 93/8; Kliphoogte (-CD), Bayer 1652 (NBG). 2917 (Springbok): Lekkersing (AA), Smithers in NBG3092/35 (BOL); Karrachabpoort (-AB), Bayer 1674 (NBG); Steinkopf (-BA), Meyer (NBG); N. Steinkopf (-BB), Venter 88/35; Springbok (-CA), Hurling & Neil (BOL); 20km E. Springbok (-CA), Salter 3773 (BOL); Kourkamma (-CD), Bruyns 6745 (NBG); Meyer in PRE 34984. 2918 (Aggenys): Witbank (-AC), Venter 92/105; Kangas (-CB) Venter 91/44. 3017 (Hondeklipbaai): Soebatsfontein (-AB), Le Roux 3991 (NBG). 3018 (Kamiesberg): Hosabees (-AA), Venter 89/37; Putsiesekloof (AD), Bruyns 4774 (BOL); Kotzesrus (-CA), Bruyns in KG 274/76 (NBG); 6km NE. Garies (-CA), Leistner 733 (PRE); NE. Bitterfontein (-CD), Venter 91/43. 3019 (Loeriesfontein): Donkiedam (-CC), Bruyns 6818. 3118 (Vanrhynsdorp): Komkans (-AA), Peers (BOL); Mierhoofdskastell (-AA), Barker 6383 (NBG), Venter 91/73; SE. Nuwerus (-AB), Lewis (BOL); NW. Nieuwoudtville (-BB), Venter 94/28; Liebendal (-CB), Hall (NBG). 3119 (Calvinia): Ezelskop (-AA), Bruyns 6832; Koringberg (-AB), Bayer 3398 (NBG); Beeswater (-BC), Bayer 3423 (NBG); Keiskieberg (-DB), Perry 1994 (NBG). 3219 (Wuppertal): Skitterykloof (-DC), Bayer 2574 (NBG); Ceres Karoo (-DD), Bayer 1619 (NBG), Herre in Stell.6391 (BOL). 3220 (Sutherland): Ganagas Pass (-AA), Venter 89/51; Ouberg Pass (-AD), Bruyns 2547 (BOL); Verlatenkloof (-DA), Bayer 4320 (NBG); Sutherland to Laingsburg (-DD) Smith 7392 (NBG).
Inadequately located: Little Namaqualand, Britten in PRE 34983.
-

Haworthia arachnoidea var. namaquensis JDV91/43 north of Bitterfontein. Invariably in the shade of bushes or stones. -

Haworthia arachnoidea var. namaquensis JDV94/24 east of Springbok. The plants are usually withdrawn into the soil like this. -

Haworthia arachnoidea var. namaquensis JDV91/155 Umdaus. This variety seems to flower in early spring.
d. var. nigricans (Haw.) Bayer comb. nov.
H. setata var. nigricans Haw., Revis. :56(1821). Type: Not preserved. Neotype (designated here): CAPE-3321(Ladismith): SW. Vanwyksdorp, Bayer 2419 (NBG): H. helmiae V.Poelln., Feddes Repert.Spec.Nov. 41:201(1937). Scott :99(1985). H. unicolor var. helmiae (V.Poelln.) Bayer : 121(1976), Bayer 59:(1982): Type: Cape, Heidelberg, Smithers in Triebn. 901, Gt Brak River, Mrs Helm in Triebn. 901. Not preserved. Lectotype (B&M): icon, (B). Epitype (designated here): CAPE‑3322 (Oudtshoorn): Schoemanspoort (‑AC), Bayer 171 (NBG). H. venteri V.Poelln., Cactus.J 8:19(1939). H. unicolor var. venteri (V.Poelln.) Bayer :166(1976). Bayer :59(1982): H. aristata sensu Scott :110(1985) Type: Cape, Barrydale Distr. Major Venter (not preserved). Lectotype (designated here): CAPE-3320 (Montagu): Barrydale to Lemoenshoek (-DC), Smith 3994 (NBG).
nigricans: blackish, for the dark hues.
1982 – The type locality for H. venteri was recorded by Smith as ” 3/4m. Barrydale‑Lemoenshoek”, although Major H. Venter’s localities are very broadly stated in publication and practically valueless. Only Smith’s notes give any better clue to these but in this case there is still no certainty. However, there is a very common species extending from Barrydale to Oudtshoorn from which this variety is clearly drawn. At Barrydale itself the plants are rather pale green with elongated sparsely setate leaves – this is regarded here as the species H. unicolor. Eastwards the plants are more robust and often glabrous, usually distinguished by particularly dark‑purplish coloration towards the leaf tips. The further eastwards one progresses the more setate the plants become until at Oudtshoorn, south of the Cango Caves, the plants are comparatively small and more softly setate (H. unicolor var. helmiae).
It is not in the least clear what the relationship is between H. unicolor and H. arachnoidea. They do not appear to cohabit which suggests that they may be conspecific and only differentiated on an ecotypic basis. This is borne out by the pale arachnoidea‑like population at the northern entrance to the Tradouw Pass, south of Barrydale. However at Ladismith H. arachnoidea is present as a very distinct heavily setate form, and similarly south of the Cango Caves it occurs near to H. unicolor var. helmiae as a softer haired but also highly setate form. The species H. unicolor is therefore not well‑known or properly understood and this solution must be regarded as suspect. Von Poellnitz’s citations of localities for the var. helmiae are highly confusing and in Feddes Repert. Spec. Nov. 44:223(1938) are totally disparate. The type is cited in 1938 as “Great Brak River, Mrs Helm” and three Triebner numbers are added to this. Mrs Helm’s strong personal recollection (private communication, and acknowledgement to Col C.L. Scott) is that the original plants were collected at a specific site south of the Cango Caves, Oudtshoorn. The photograph extant in the Botanical Museum Dahlem (which must serve as the type) is of a very poor specimen, but it can be reasonably allied with plants from the site given by Mrs Helm. It is concluded that it is an extension of the complex to which it is here referred. J.R. Brown’s illustrations in Cact.Succ.J(U.S.) 18:39(1946) are not of this variety.
1999 – The incorporation of H. unicolor var. venteri under H. arachnoidea as the var. nigricans of Haworth represents a major change. Smith does state that his collection is from the locality to which he was directed by Col Venter. The type selected by Breuer and Metzing for the name venteri, is doubtful because of the locality cited. The initial problem was to relate unicolor (= H. mucronata) to a substantial species body, which I could not previously do. How this problem is resolved is also discussed under H. mucronata. It is clearer now that var. nigricans is better associated along its northern and western borders with var. arachnoidea and there are areas where it is not possible to make a distinction between these two varieties. This realisation suggests the re-application of Haworth’s original name which means blackish-green. From records of Dekenah’s collections it appears that var. venteri does interface with H. mucronata in the southwest and I can confirm this from my own collection from south of Vanwyksdorp.
Regarding H. helmiae: neither of the three numbers given by von Poellnitz in 1938 agrees with Heidelberg, Smithers in Triebner 901 as in the original description, to which was added a collection of Mrs Helm’s from Great Brak. In addition Von Poellnitz cites several quite disparate and improbable localities between Worcester in the west and Avontuur in the east. Where H. helmiae was reputed to have been actually collected (Schoemanspoort), fairly glabrous forms occur together with softly spinose ones. These are small dark elements with very reduced spines and often without spines at all (they do not resemble the softish translucent plants figured by Brown as mentioned above). They must be closely associated with the var. nigricans (previously unicolor var. venteri) and the change of interpretation here makes it very much easier to accommodate a very wide range of forms which tend to be glabrous and without any spines.
Southwest of Oudtshoorn var. nigricans can be robust with heavy spines and it is probably here that H. ferox var. armata V.Poelln. originated. The point is made that H. arachnoidea does have glabrous variants and they are generally accommodated in this variety.
It is distinguished by the purplish coloration towards the tips of the leaves and the generally darker colour. The leaves tend to be broader and the spines larger and sparser than for the species generally. Glabrous forms with pronounced keels commonly occur and the more delicate softer versions of this can perhaps be ascribed to H. mucronata var. integra. These variants of H. mucronata are distinguished by their translucent margins and keel. The interaction between that species and H. arachnoidea is an interesting field for investigation.
Distribution: 3320 (Montagu): Jagerskraal (-AB), Bayer 3612 (NBG); Kareevlakte (-AD), Archer (BOL); SE. Konstabel Stn.(-BC), Bruyns 2448 (NBG); Elandskloof (-BD), Bruyns 2418 (NBG); Prinspoort (-BC), Venter 91/114 (NBG); Keurfontein (-BD), Bruyns in KG18/76 (NBG); 24km Montagu to Ladismith (-CB), Esterhuysen (BOL); Bellair Dam (-DA), Venter 5 (NBG); E. Barrydale (-DC), Bayer 4671 (NBG), Stanford (BOL); W, Barrydale (-DC), Smith 5770, 7310 (NBG); Brandrivier (-DD), Smith 3995 (NBG); Near Barrydale (-DD), Smith 5672, 3994 (NBG); E. Lemoenshoek (-DD), Hurling & Neil (BOL), Bayer in KG126/72 (NBG), Stayner in KG15/67 (NBG); Warmwaterberg (-DD), Bayer 1704 (NBG); W. Warmwaterberg (-DD), Bayer 4575 (NBG). 3321 (Ladismith): N. Calitzdorp (-BC), Bayer in KG441/75 (NBG); Winkelplaas (-CA), Bayer 2716 (NBG); S. Ladismith (-CA), Smith 5506 (NBG); Ockertskraal (-CA), Smith 4004, 5510 (NBG); W. Ladismith (-CA), Smith 7140 (NBG), Bayer 1624 (NBG); Die Eike (-CA), Laidler 314 (NBG); SW. Ladismith (-CA), Bayer 1615 (NBG); Kareebosch (-CA), Laidler 481 (NBG); Adamskraal (-CA), Smith 4002 (NBG), Bayer 1617 (NBG), Ferguson 5 (BOL); Ladismith to Vanwyksdorp (CB), Bayer in KG573/51 (NBG); Springfontein (-CC), Smith 5657, 7325 (NBG), Bayer in KG 166/71 (NBG); W. Springfontein (-CC), Smith 5379 (NBG), Bayer in KG167/71 (NBG); Muiskraal (-CC), Smith 3989, 7136 (NBG), Bohnen 8416 (NBG); Riversdale to Ladismith (-CC), Smith 537 (NBG); Muurkeeskraal (-CC), Smith 4000, 3976, 3995, 3997, 7138 (NBG); W. Vanwyksdorp (-CD), Bayer 2418 (NBG), Stayner in KG57/67 (NBG); S. Vanwyksdorp (-CD), Bayer 2419 (NBG); S. Calitzdorp (-DA), Bayer in KG126/72 (NBG), Bayer in KG125/73 (NBG); Oudtshoorn to Calitzdorp (-DB), Bayer 1616 (NBG); Warmwaterbron (-DB), Bayer in KG115/71 (NBG). 3322 (Oudtshoorn): Schoemanspoort (‑AC), Bayer 171 (NBG); N. Oudtshoorn (-AC), Bayer 4499 (NBG). Volmoed (-CA), Bayer 4656 (NBG), Zeekoeigat (-CA), Gie (NBG); Mt Hope (-CB), Bruyns in KG437/75 (NBG).
Inadequately located: Near Doornrivier, Helm in Fourcade 4740 (BOL); Caledon, Venter 9 (BOL);
-

Haworthia arachnoidea var. nigricans JDV south of Calitzdorp. In close proximity to H. mucronata var. rycroftiana, -

Haworthia arachnoidea var. nigricans JDV92/52 west of Ladismith. This area has produced the var. setata and H. mucronata var. inconfluens. -

Haworthia arachnoidea var. nigricans JDV94/31 west of Muiskraal. Glabrous forms grow intermingled with setate forms. -

Haworthia arachnoidea var. nigricans JDV89/57 Anysberg. This is one of the few places where the var. setata and H. mucronata are also present.
e. var. scabrispina var.nov.
Type: CAPE-3320(Laingsburg): Baviaans (-BA), Bayer 2105 (NBG. Holo.).
scabrispina: rough-spined.
Rosette roundish and raised above ground level. Leaves with firm rigid brownish spines. (A var. arachnoidea spinis rigidis bruneolis differt).
The var. scabrispina is created to accommodate the southern Karoo elements which have very hard stiff spines. The plants tend to be raised and the leaves and spines are fairly rigid so that the plants form quite rounded spheres rather than flattened rosettes. There are populations in which the plants have the spines produced from translucent raised bases and also in which the upper petals do not curve upward. This seems to suggest some affinity with H. marumiana var. archeri.
Distribution: 3220 (Sutherland); 35km N. Laingsburg (-DC), Bayer 2124 (NBG). 3221 (Merweville): Schoppelmaaikraal (-CD), Bruyns in KG30/76 (NBG). 3320 (Montagu): N. Baviaans Stn. (-BA), Bayer 2105 (NBG); Whitehill Ridge (-BA), Barker 13368 (NBG), Archer (BOL); Laingsburg (-BB), Lewis & Barker in NBG2772/32 (BOL); Nougaspoort (-CA), Bayer 1963 (NBG); 20km W. Ladismith (-CA), Bayer 4462 (NBG).
-

Haworthia arachnoidea var. scabrispina JDV91/111 Rooinekpas. There are several collections of this dark form which has similar flowers to H. marumiana var. dimorpha. -

Haworthia arachnoidea var. scabrispina JDV89/59 south-west of Lainsburg. Grows very near to H. nortieri var. pehlemanniae, but is usually on shadier south-facing slopes. -

Haworthia arachnoidea var. scabrispina JDV95/31 south-east of Laingsburg. With very rounded rosettes and brown end-awns.
f. var. setata (Haw.) Bayer comb. nov.
H. setata Haw., Suppl. Pl.Succ.:52(1819). Brown, Cact.Succ.J(U.S.) 16:3(1944). Scott, Aloe 16:42(1978). Scott :68(1985). Type: not preserved. Neotype (see Scott, 1985): icon, artist unknown, specimen received from Dr Mackrill ex Cape, (K): H. setata var. media Haw., Revis. :56(1821). Type: Not preserved: H. setata var. major Haw. ibid. Type: Not preserved: H. gigas V.Poelln., Feddes Repert.Spec.Nov. 31:84(1932). H. setata var. gigas ibid. 44:224(1938). Type: Cape, Amalienstein, Stellenbosch 6692 (not preserved). Neotype (designated here): CAPE-3321(Ladismith): Amalienstein, Smith 7390 (NBG): H. minima var. major V. Poelln., Kakt.u.and.Sukk. :39(1938). H. tenera var. major Uitew., Sukkulenta :52(1948). Type (designated here): Cape, Derust, Mrs Helm (not preserved). Neotype: CAPE-3322(Oudtshoorn): Meiringspoort (-BC), Bayer in KG 160/72 (NBG): Aloe setosa Roem. et Schultes, Syst.Veg. 7:641(1829). Salm-Dyck, Monogr. 12:f3(1841). Type: Not preserved. Neotype (designated here): icon, f3 Salm Dyck, Monogr. 12(1840).
setata: bristled.
This variety tends to have dense white spines which do not blacken on drying, and the texture of the species is always softer than the var. scabrispina. It is quite a variable element and like the var. nigricans is also closely interwoven with H. mucronata. Although usually moderate in size and only upto about 8cm in diam., some robust specimens occur east of Ladismith. Two specimens are cited below from the Jansenville district and it is more probable that they belong with H. decipiens var. cyanea.
Distribution: 3224 (Graaff Reinett): Jansenville (-DC), Long 1321 (PRE); Jansenville (-DC), Marloth 9384 (PRE). 3320 (Montagu): Keurkloof (-BC), Barker 1913 (NBG); Elandskloof (-BD), Bruyns 2259 (NBG); Buffelsrivier Pass (-BD), Bruyns 2564 (NBG); Cogmans Kloof (-CA), Esterhuysen (BOL), Hurling & Neil (BOL), Long 1124 (PRE), Smith 7224 (NBG), Stayner in KG38/67 (NBG), Otzen in NBG 1316/35, Barker 8266 (NBG), Smith 3989, 7490 (NBG); Prins River (-DB), Bruyns 2462 (NBG); Barrydale (-DC), Smithers (BOL); Tradouw Pass (-DC), Hurling & Neil (BOL), Smith 3991, 6782, 6789 (NBG), Thompson 664 (NBG). 3321 (Ladismith): Amalienstein (-AD), Herre in Stell.6639 (BOL), Smith 7390 (NBG); Huis River Pass (-BC), Smith 7389 (NBG); Opsoek (-BC), Smith 6886 (NBG); S. Ladismith (-CB), Bayer in KG 160/72 (NBG), Smith 5507 (NBG), Joubert 10 (BOL); 5km S. Ladismith (-CB), Smith 5652 (NBG); 6km S. Ladismith (-CB), Smith 5729, 5733 (NBG), Stayner in KG42/70 (NBG); 14km E. Ladismith (-CB), Bayer in KG593/71 (NBG); W. Vanwyksdorp (-CB), Bayer in KG535/71 (NBG); Groenfontein (-DA), Smith 6888 (NBG); Calitzdorp (-DA), Fouche 53 (PRE); E. Vanwyksdorp (-DA), Bayer 5753, in KG388/71 (NBG). 3322 (Oudtshoorn): 32km S. Prince Albert (-AC), James (BOL), Taute (BOL); Boomplaas (-AC), Moffett 141 (NBG); Schoemanspoort (-AC), Bruyns in KG92/77 (NBG), Smith 5230 (NBG); Meiringspoort (-BC), Bayer in KG160/72 (NBG); Oudtshoorn (-CA), Smith 5785 (NBG), Bolus 12375 (BOL); N. Dysselsdorp (-CB), Bayer & Venter 6608 (NBG). 3324 (Steytlerville): N. Steytlerville (-AC), Bayer & Venter 6610 (NBG).
Inadequately located: Ladismith, Jordaan (BOL), Joubert 11 (BOL); Sundays River Estate, Heatlie in NBG254/25 (BOL).
-

Haworthia arachnoidea var. setata JDV94/86 Dyseldorp. There are also populations of vr. aranea in the area. -

Haworthia arachnoidea var. setata JDV93/23 east of Steytlerville. A population north of Steytlerville flowers in September at the same time as the var. aranea. Considered to be tending towards the var. xiphiophylla which also flowers early. -

Haworthia arachnoidea var. setata JDV92/93 east of Anysberg. Co-occuring with the var. nigricans. It does not have the purplish tips to the leaves. -

Haworthia arachnoidea var. setata JDV90/70 De Rust. There are several populations like this which tend towards the var. aranea. -

Haworthia arachnoidea var. setata JDV92/100 south-west of Ladismith. This very white-spined form is included in the var. setata.
g. var. xiphiophylla (Baker) Bayer comb.nov.
Haworthia xiphiophylla Baker, Fl.Cap. 6:354(1896). Baker, Curtis’ Bot.Mag. t.7505(1896). Bayer :168(1976). Bayer :61(1982). Scott :73(1985). H. setata var. xiphiophylla (Bak.) V.Poelln., Feddes Repert.Spec.Nov. 44:225(1938). Type: Cape, Uitenhage, Howlett (K): H. longiaristata V. Poelln., Kakteenkunde 9:134(1937). Type: Not preserved. Neotype (designated here): CAPE-3325 (Port Elizabeth): N. Coega Station (-DA), Mrs E.B. King (NBG).
xiphiophylla: sword‑like leaves.
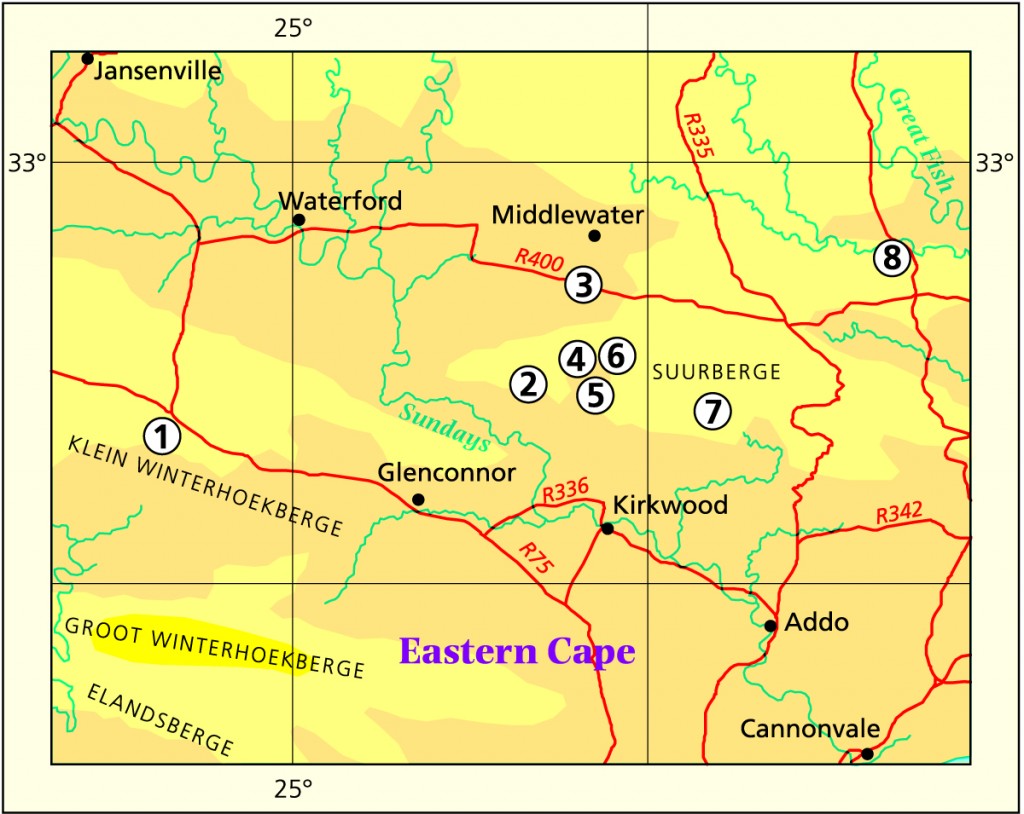
1982 – This is a bright green species with broad but short, well‑spaced marginal teeth. The leaves are long and slender. H. xiphiophylla is found in the vicinity of Uitenhage and Coega. It has been regarded as a variety of H. arachnoidea but this is highly unlikely from the viewpoint of distribution only. A number of anomalous populations at Coega, Dead Man’s Gulch and Kirkwood confuse the issue. Thus H. translucens and H. xiphiophylla pose an intriguing and interesting problem for the field worker.
1999 – The latter sentence is also fairly typical of the loose words used when the herbarium record is inadequate. This is a bright green variety with broad but short, well‑spaced marginal teeth. The leaves are long and slender. The incorporation into H. arachnoidea is to include the variables along the southern Swartberg mountains from Anysberg through to Steytlerville where the species meets with the Eastern Cape variants. One of the populations alluded to is represented in this work by the ‘re-discovered’ H. aristata. From the collections by J.D. Venter, it seems that var. xiphiophylla does merge directly with var. arachnoidea at its westerly limits. I would have expected a relationship with the greener, braoder leaved H. decipiens var pringlei as discussed under that species. However, var. xiphiophylla does not develop translucent windows and the leaf spines tend to be separate and narrow at the base. The flowering time appears to be early summer ahead of typical var. arachnoidea which flowers in the summer.
Distribution: 3324 (Steytlerville): 30km E. Steytlerville (-BC), Venter 85/68 (NBG). 3325(Port Elizabeth): Mentz Dam (-AA), Venter 91/122 (NBG); S. Mentz Dam (-AC), Venter 91/117 (NBG); Mannetjie (-CA), Venter 94/19 (NBG); Maraishoop (-CA), Smith 3583 (NBG); Bauerskraal (-CB), Venter 93/75 (NBG); Sandfontein (-CB), Archibald in NBG 1326/32; Uitenhage (-CB), Britten in PRE 34951; De Rust, 2km NW. Couga Kop (-DC), Branch 5 (NBG); Couga Kop (-DC), Smith 3546 (NBG), Britten (BOL).
-

Haworthia arachnoidea var. xiophiophylla JDV93/75 north-west of Uitenhage. Longer more slender leaves and relatively larger and more widely spaced spines. -

Haworthia arachnoidea var. xiophiophylla JDV96/88 west of Coega. The plants are seldom solitary like this and dense stands may occur. -

Haworthia arachnoidea var. xiophiophylla JDV94/98 Coega. The leaves are generally more yellow-green and the spines shorter and broader than in the other varieties.
[ed.] – Also see A fleeting look at Haworthia arachnoidea, and A shadow of the past – Haworthia arachnoidea again. (1999)