A supposed new species of Haworthia viz. H. groenewaldii Breuer, is described in an article authored in Alsterworthia 11.2:13-17 by Breuer, Marx and Groenewald. It presents the description of this supposed new species from Buffeljags east of Swellendam that I would simply have identified as another variant of H. mutica. This is not because I am confounded by the variability among the plants in the genus, or even in any one species. I recognize the species as systems of populations in which the individuals vary from one another as one would expect in any group of living things that maintains the flexibility to adapt to constantly changing world conditions. In this even time becomes a variable. I think species are very important elements if the whole of creation and not just for taxonomist and collector activity. Other people have other ideas of what species are, so my disagreement is hopefully forgivable..
Although H. mutica was described by Haworth in 1821 it was not allied to a South African field population until recognized by Col. Scott in 1985. G.G. Smith had failed to recognize it when he described his H. otzenii in 1945. The type by which the name is supported is a Kew illustration reproduced here as Fig. 1. This then is what one would expect a typical representative of the species to look alike. Now the ever present problem in Haworthia, is that no two plants in a population may look quite the same. Hence my problem with the description in which the word “typical” is rather bandied about. It falls into the first aspect of taxonomy.

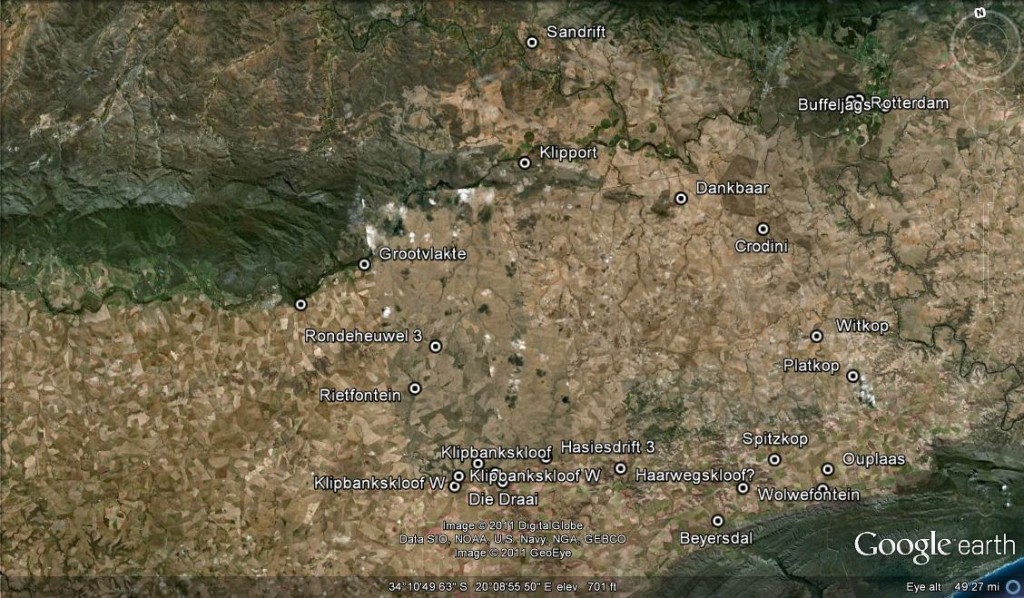
Firstly a plant is illustrated on the front cover of the respective Alsterworthia that, presumably the authors, state is a typical specimen of H. groenewaldii. Secondly, Marx is quoted as saying that the “typical H. mutica” grows only 20km west at the farm Dankbaar. Statements like these are used to strengthen and support opinions and generate a reality that Latin binomials sadly lack. It so happens that I know both these populations quite well and these statements are news to me. I do not think the specimen on the front cover is by any means typical of the population at Buffeljags, and certainly not at three sites recently discovered nearby. The plants at Dankbaar also do not in my opinion fully meet the imputed similarity to fig.1. See fig 2. for an image of a plant representative of the Dankbaar population. I would be very hesitant to say that this is typical of Dankbaar plants.

There is a curious problem here in that Scott does not use the type illustration in his Revision and does not state any origin of the plant he uses to illustrate the species i.e. H. mutica. As far as I am aware the type of a synonym that G.G. Smith described viz. H. otzenii, came from east of Riviersonderend, but this is for an Otzen collection no. 6. The type was cited by Scott as Otzen 10 but this is not in the Compton herbarium where it should be. So I am not sure offhand where that came from. However, this is not really relevant to the discussion. I just want to state that finding a plant that matches the type is no mean feat and that it was by sheer chance that in 1969 I came across a population of plants at Hasiesdrift that did. See fig. 4 and 5. I selected one image and then realized that it did not have round enough leaf tips to meet need, so I selected another. In the first picture the leaves tend to have a “mucro” – a small point to the leaf that looks different to a well developed end-awn (bristle) that the leaves can also have.



Another issue is that Gerhard Marx once argued with me that the mooted H. groenewaldii was much nearer to H. mirabilis than to H. mutica as I had suggested. What he luckily is able to ignore is my observation of the similarity of some plants of H. mutica to H. mirabilis see figs 5 and 6. I never saw a plant quite like fig. 5 in all my exploration at Buffeljags but Jannie Groenewald collected this one there. Fig. 6 is not typical of the population either and I used this same figure somewhere else in my writing to comment on the similarity to H. mirabilis ‘badia’ variants at Sandfontein (east of the “typical”). Gerhard is still more fortunate to be able to ignore the similarity of H. mutica to H. retusa see figs 7 and 8. I even surprised myself when in looking for a suitable picture, from many, I picked this fig. 7 and find it is also Hasiesdrift albeit a cultivated version. (There is such an interesting story around the Hasiesdrift site). Fig. 8 is a representive of H. retusa from Pienaarsriver pictures and I feel sure that readers will agree that the names could be switched. It was very difficult to ignore pictures from Pienaarsriver that I could have used with figs 5 and 6 in the context of H. mirabilis.



It becomes still more interesting (I would have said complicated but my critics maintain that this variation confuses me and it is actually possible to get it all tidy and neat) when one further compares a plant of H. retusa ‘nigra’ ( fig. 9) with both H. mutica (fig. 7); and then H. retusa ‘nigra’ (fig. 10) with H. retusa (fig. 11). The latter is in fact from the population where the variant ‘geraldii’ originates.



A last point I can make is that the leaf flecks said to characterize “H. groenewaldii” do occur in H. mutica at Klipport (see figs 12 & 13). One cannot simply ignore the extraordinary chain of similarities that extends across the entire distribution area producing only a slightly different situation at either extreme.


Floral difference is a great issue that is misused to force an opinion. The flower is extremely difficult to study because the differences across the entire subgenus are so small. There are complications where, as an example, flowers of H. pulchella ‘globifera’ are indistinguishable from those of H. cymbifomis var. incurvula. In the subg. Hexangulares there is an incredible problem where floral differences within species exceeds that between species e.g. a flower of a plant of H. limifolia may more closely resemble that of H. coarctata rather than that of another plant of H. limifolia. It is easy to draw conclusions from small samples of a few flowers from a few populations but one very soon finds that with increasing sample size the most carefully constructed table of differences becomes senseless. Just when is an ever-aging flower on a stalk at the precise same stage of a flower you want to compare it with? How many flowers from how many plants are needed to make a valid statement? Do not forget that the observations must be made on plants on the basis of random selection too. This is a requirement mostly totally ignored when the more serious question of a species difference is being debated. That of course brings us back to this casual use of the word “typical” and its intractability.
The hardest problem to deal with is that of flowering time and on the face of it a winter flowering time versus a summer flowering time can be taken to suggest significant difference. Yet if one considers that a species needs to harbour genetic variation to ensure adaptation to any kind of environmental change and so survival, a different flowering time may be an extremely useful resource. Then we do have the reality of hybrids between species that do flower at these different times. So obviously and self-evidently populations of species may exist that has arisen from such hybridization between plants that flower at different seasons. The capacity of plants and animals to synchronize breeding periodicity is well-known.
I close with my observation that H. mutica is an assorted group of plants that seem to fall in some middle zone between H. mirabilis and H. retusa. Hence in the west we have H. mirabilis/ H. mutica/H. retusa ‘nigra’, while in the east we have H. pygmaea (=H. mirabilis+H.retusa ‘retusa’) and H. retusa ‘turgida’. In the area between there is vast variation of H. mirabilis and H. retusa that get a bit of H. floribunda thrown in too.
So am I confused, or have I confused you? As Steven kindly put it…’it’s almost as if you were being blamed for nature’s complexities’. Of course the ultimate reality is that we each have our own idea of what “species” are, and here I have used my version! ♦

Pingback: Haworthia mutica (groenewaldii) and its twisted leaves. Haworthia Updates