Previously published Cact. Succ. J. (Los Angeles) 84(1): 41-50.
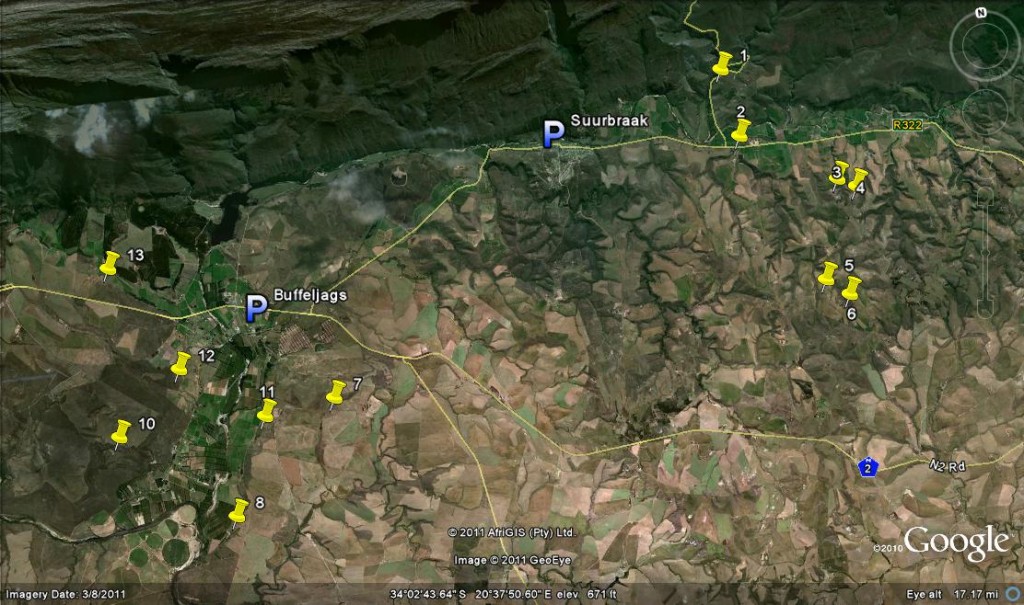
Map Legend – east of Swellendam.
1. JDV84/75 Haworthia retusa ‘turgida’.
2. MBB6666 H. retusa ‘nigra’↔ H. mirabilis.
3. MBB7898 H. retusa ‘nigra’.
4. MBB7899 H. retusa ‘nigra’.
5. MBB7897 H. retusa ‘nigra’.
6. MBB7896 H. retusa ‘nigra’.
7. MBB7871 H. mirabilis.
8. MBB7823 H. mirabilis.
9. MBB7909 H. mirabilis
10. MBB7805 H. mirabilis.
11. MBB7801 H. mutica ‘groenewaldii’.
12. MBB7886-7889 H. mutica ‘groenewaldii’, H. mirabilis, H. minima, H. marginata.
13. MBB7722 H. floribunda ‘major’
1. Haworthia marginata and H. minima
The Robustipedunculares is quite a distinctive group within the currently recognized genus. While the four species in the group are generally quite distinct, there are some remarkable complexities that rival that elsewhere in the genus. Recognizable and obvious hybrids are found between H. marginata, H. pumila and H. minima, but there are instances of whole populations that appear to consist of such in-between forms e.g. H. Xmortonii. I have speculated that perhaps rather than have just hybridized, the species have never ever really truly separated in the supposed evolutionary process. There is a vast body of variants that still link them in intimately as this piece will show.
I long ago observed that a small remnant population of ostensibly H. minima just south of Swellendam flowered in November as opposed to the general rule for the Robustupedunculares as late summer flowering. A vicarious population at Brandrivier north of the Langeberg (H. minima ‘opalina’) also flowers in November and both populations have fairly large and white flowers for the species.
I have recorded the normal bluish-green H. minima within the Bontebok Park at Swellendam as well as a very green variant. But in very recent exploration to the south-east we found an even more divergent group of plants that, while varying among the plants, seemed to be hybrids of H. marginata and H. minima (fig. 2 as a single sample and not representative of all). It was September and there were no signs of old or new flower spikes. Kobus Venter, who was present, remarked that the first plant seen was reminiscent of the plants once present south of Swellendam. The plants were large and in exposed situations even colored brownish as does H. pumila. No flowers were present and their color may have shown if H. pumila could have been directly involved at all, while it is essentially its distribution restricted eastwards from the Robertson Karoo by some 20km that reduce the possibility.
What makes the situation more interesting is that nearby was a population of H. marginata that was flowering and the flowers were also large and very white for the species (figs 3 & 4). Added to the fun was a smaller probable hybrid (fig. 5).
Differences and complexities like this do not really surprise me because it is what I have come to expect in my many wanderings in the field. The problem is that it certainly makes classification and any agreement on a set of names very difficult. I just accept it as a fact that plant species can exhibit greater differences between individuals of the same species than between individuals of different species, ridiculous as it may sound. This is because I perceive species as systems of individuals in populations with a very strong geographic component. To actually make a decision it is frequently necessary to determine just what else is growing in the vicinity in respect not only of the genus in question, but also that of other plants. Even the habitat factors need to be considered.
In the case of the plants pictured with this, the Bontebok Park terrain is mostly tertiary gravels, while the habitat we found the plants in was more recent riverine boulders. It is very curious that in the description of H. groenewaldii, the habitat is implicitly described as Ruens Silcrete. I do not think this is true. It is in the true Ferricrete – Silcrete that we found the next and it seems to be these differences in substrate geology that play a large role in generating variation and consequent controversy.




2. Haworthia retusa ‘nigra’
I first allied this element with H. mutica because this is how G.G. Smith referred to his collection from Kransriviermond south of Heidelberg. Since then there have been many new collections from which can be gathered that this population is a hybrid one between H. retusa ‘turgida’ and H. mirabilis. There is another collection north of this at Morning Star that appears to have the same parentage but also including H. floribunda. Then there are populations continuing up the Duiwenhoks and then Klip rivers to northwest of Heidelberg, a population between Heidelberg and Tradouw Pass further west and also a population at Goedverwagting south from there. Apart from the Morning Star population (February – March) these are all September/October flowering. There is a population at the southern entrance to Tradouw Pass of the same ilk that is February/March flowering. Then there is quite a distance between these known populations and a remarkable population at Buffeljags south of Tradouw Pass. It is remarkable for the fact that it is lauded as a new species viz. H. groenewaldii when I consider it to be generated from the interaction of H. mirabilis, H. mutica and H. floribunda. I attach no special importance to the fact that it flowers, contrary to H. mutica, in February – March. This is because I have observed many hybrids between patently different species despite a seasonal difference in flowering time.
To explore the realities of the situation we undertook two expeditions, one was to Buffeljags to explore west of H. groenewaldii and the other was to the Tradouw Pass area to explore H. retusa ‘nigra’. The first exploration yielded three populations of ‘groenewaldii’, which convince me that despite its flowering time as for H. marginata above, is simply H. mutica in another guise. I also think far too much is made of superficial and trivial differences that are as much characteristic of the variation in the one original population as they are within the four populations and for H. mutuca in its full sense. I consider it significant that H. mirabilis in its more normal non-retuse and dark green form is present in discrete populations both at and west of Buffeljags.
The second expedition was nearly as fruitful. It showed the Tradouw Pass population to be February/March flowering (see figs 6 & 7), while three new populations we discovered between there and the previous records in the easterly Heidelberg direction were September – October flowering. They link up to the populations elsewhere that I assign to H. retusa ‘nigra’. An additional find by Jannie Groenewald, for whom that H. mutica viz. H. groenewaldii, variant is named, also took us to a population of what is clearly H. mirabilis (see map) as I know it in its many disguises in the white kaolinic/bentonite clays of the silcrete – ferricrete inselbergs throughout the low-lying Southern Cape. There is unquestionably an overlap of characters between what I assign to H. mirabilis and H. retusa and I consider inarguable that H. mutica is a reflection of a shared gene pool.


What this demonstrates again, as does the Kiewietsvlakte populations between Heidelberg and Riversdale, that H. retusa and H. mirabilis are closely intertwined from east to west. There is an added complexity that H. floribunda is admixed along the northern populations and H. variegata along the southern. The admixture of the two species produces H. retusa ‘turgida’ and H. pygmaea in the east and H. mirabilis, H. mutica and H. retusa ‘turgida’ in the west. This is complicated by the other interactions along the northern and southern areas. In the Potberg area it appears that the genetic material of all five “species” is evident in the populations that I have seen there.
While I would like to explain the situation around H. groenewaldii I. Breuer that I interpret as a variant of H. mutica, this should be left for another occasion as too many images are required to support any argument. As it is, the issue of H. mutica ‘nigra’ occupies 29 pages and 79 illustrations in my book Haworthia Update Vol.2 pt 1:50, where all the above mentioned variants are discussed and illustrated. The naming of Haworthia is highly contentious because the species consist of aggregates of small fairly isolated populations that may differ to large or small degree. The populations are in turn also aggregates of plants that can all be identical from vegetative propagation, similar because of low genetic difference or very different from each other because of large genetic differences. Therefore figures 6-13 simply show just a sample of the variation within these four populations in the Tradouw Pass area. The plants vary quite considerably in size too and the one in Fig 8 is nearly 200mm diameter. I have added to the map locations of the only significant other populations that I know of in the area including H. retusa ‘turgida’ and H. floribunda ‘major’, excluding those within the Bontebok Park viz. H. minima, H. mirabilis and H. marginata.







Perhaps I should close by explaining that I have dropped the use of any rank below that of the species name. I simply am suggesting that we recognize the need for a trinomial system without the pretension of status, and more greatly honour the binomial as an entity of a greater significance than we may know. I do this because botany has no proper species definition and consequently species descriptions are just based on wild guesses about possible non-similarity and on the flimsiest of supposed character differences. The loosely used word “typical” is only truly useful in respect of the one plant dried as an almost unrecognizable specimen that is used to anchor the Latin name.
Acknowledgement. Any proper excursion into Haworthia territory always requires acknowledgement of landowners and I thank Jaap Viljoen and Jannie Groenewald for organizing that and for their company. I was also glad to have Kobus Venter along who had persuaded me to show him some of populations known to me on promise of new exploration.
Note.
Cross seasonal hybrids observed are-
H. retusa turgida X H. floribunda Blackdown, Heidelberg.
And also Platjieskop, Riversdale.
H. pygmaea X H. floribunda Coopert Siding, Albertinia.
H. mirabilis X H. retusa Soetmelksrivier, Riversdale
H. mirabilis X H. variegata Stoffelsriver, Swellendam.
References. I need to record that Harry Mays of Alsterworthia kindly undertook the non-profitable publication of 5 volumes of Updates (2-6) between 2006 and 2009. Vol. 1 was published by Umdaus in 2001. These volumes were the product of research to validate or correct what appeared as a formal revision in Haworthia Revisited, published by Umdaus in 1999. The description of H. groenewaldii appears in Alsterworthia 11.2:15 (2011). ♦