2. THE RACEME. Figures Set 2 show the bases of the peduncles in several collections to again show how variable they are and not only because of plant vigor and current growth conditions. Diameter can vary by a factor of three. Color is variable and the bract spacing and size of bracts as variables must be noted. Fig 6 is simply a robust spike in a population where the flowers were sparsely spaced on the stem with approximately 15-20 flowers per stem, whereas this raceme had nearly 30 flowers. The number can drop to as low as three. In 7917 we noted a single plant with an inflorescence of over 600mm where the average was below 300. Similarly at 7818 there was one colossal inflorescence of 800mm where again the average was between 300 and 400mm. There is a real problem in that the typological attitude is often adopted when making comparisons like this. An extreme example would be to take H. retusa south of Riversdale as typical of the species. These are massive plants (source of ‘Jolly Green Giant’) and the inflorescences are huge with many flowers. This is not typical for the species and especially so if one takes the mountain cliff populations (H. turgida) to be the same species (as I do) where the plants are proliferous and the inflorescences many and reduced. Plants in poor niches and even poor habitats, flower weakly and the inflorescences are reduced. Figs 7 shows varying capsule positions on the stem. Figs 8 and 9 show a distichous and a secund inflorescence and figs 10 to 15 demonstrate the varied spacing of the flowers that is observable even in any one population although the images are for two different accessions. In Fig. 8 the middle flowers are in a single plane and I regret not having observed the leaf arrangement in that specific plant, because this is a distichous arrangement as the low Fibonacci number of a spiral arrangement of the flowers. This may have been reflected in the leaf arrangement too. The spacing and arrangement of the flowers is also a variable and the number of flowers may also vary. Figs 16 to 18 show bud arrangement and the way in which the fish-tail buds have upturned tips. Although the peduncle does continue to lengthen, most of the lengthening takes place in the flower producing area and towards the upper end of the raceme. The peduncle does not always stay straight and may bend slightly at each floret. The number and distance of the flowers along the peduncle may affect bud packing just as peduncle formation in the rosette results in the appearance of a groove on the leaf face. This is a secondary physical phenomenon and is not an inherent “character”. The leaf keel for example may also be influenced just by physical leaf-packing. A peduncle of a flower from the centre of a plant will have a many angled base, but one arising from a leaf axel only 2-angled. Generally the raceme is indeterminate, meaning that it does not end in a pedicel and flower. But I have seen an individual raceme of H. floribunda with a terminal flower. This exemplifies our problem where characters one might think could be used to determine even genera, are variables at species level.



2.1 7075 H. mutica, Grootvlakte
2.2 7262 H. mirabilis, S Greyton
2.3 7955 H. mirabilis, Van Reenens Crest


2.4 7778 H. mirabilis, Komserant
2.5 7809 H. mirabilis, Koeisekop




2.6 7913 H. mirabilis, Rietkuil
2.7 7919 H. mirabilis, Van Reenens Crest
2.8 7918 H. mirabilis, Van Reenens Crest
2.9 6509 H. mirabilis, W Bonnievale



2.10 7809 H. mirabilis, Koeisekop
2.11 ADH2729 H. mutica, De Hoop
2.12 7959 H. mirabilis, W Van Reenens Crest



2.13 7075 H. mutica, Grootvlakte
2.14 7741 H. mutica, Dankbaar
2.15 7920 H. retusa ‘nigra’, Van Reenens Crest




2.16 7781 H. retusa ‘fouchei’, Komserante
2.17 7778 H. mirabilis, Komserante
2.18 7818 H. mirabilis, Windsor
3. THE BRACTS. Figures Set 3. Bracts occur from the very base of the peduncle and subtend each flower. The bracts vary in density and size and one should recall J.R. Brown’s complaint that the plant he illustrated as H. schuldtiana var. major could not be that variety because the bracts were not the same length specified by G.G. Smith in the description. Although the bracts are attached to the stem, sometimes the stem is slightly swollen as though to form a base for the pedicel and occasionally the bract seems to be attached to that as though recaulescent (Fig.3.1). Usually there is a short pedicel but these do differ in length and width (figs 6 & 7, 8 & 9) within populations so that the florets may sit against the stem and in others away from it on a pedicel 3-4mm long (figs 2&3, figs 3,4 & 5). This is again an example of what might have been a primary character i.e. stipitate vs pedicellate, separating species, as a variable within a species. The bract may be quite narrow but may also be broad and almost encircle the stem (figs 12 & 13). The margins of the bracts may be entire or serrated in the same population and other oddities do occur (figs 8 & 9).



3.1 7262 H. mirabilis, S Greyton
3.2 7818 H. mirabilis, Windsor
3.3 7818 H. mirabilis, Windsor
Bract recaulescence? Note bract positions.



3.4 7918 H. mirabilis, Windsor
3.5 7918 H. mirabilis, Windsor
3.6 7919 H. mirabilis, Van Reenens Crest
Note presence or absence of pedicel.



3.7 7919 H. mirabilis, Van Reenens Crest
3.8 7919 H. mirabilis, Van Reenens Crest
3.9 7963 H. floribunda, Niekerkshek
Double bract



3.10 7818 H. mirabilis, Windsor
3.11 7818 H. mirabilis, Windsor
3.12 7778 H. mirabilis, Komserante
Variation in bract size. Bract almost wrapping peduncle.

3.13 7960 H. mirabilis, Kruiskloof Bract tucked under flower
4. THE BUDS. Figures Set 4. These figures illustrate the bud-tip as it occurs in most of the south-western Cape species. This flattened split tip is most evident in H. herbacea and H. reticulata but also present in H. maculata, H. retusa, H. mirabilis, H. mutica and H. pygmaea, but not in H. floribunda, H. rossouwii and H. variegata. The bud also is often slightly double-arched, bending first downward and the upward again especially evident in the bud of H. mirabilis 7248 Ballyfar. However, this is not quite an absolute and in some buds the character is not clearly flattened or may even be a bit rounded. I am not confident that my observations are entirely correct as I dismissed any floral characters quite early in my exploration when it was apparent to me that using a geographical approach might first tell me something about species from which I could then infer importance of characters of any kind. The effect of the flattened tip is to compress the tips of the upper petals, but in H. herbacea and H. reticulata the petal tips are quite spreading, whereas in H. mirabilis they tend to align vertically even folding behind the upper inner.
SET 4–BUDS
Subset 1. Haworthia mirabilis – eastern zone, Riversdale area (cf. H. magnifica).



7818 H. mirabilis, Windsor, SE Riversdale. (3)



7809 H. mirabilis, Koeisekop, Heidelberg. (3)
Subset 2. H. mirabilis – central zone, Swellendam area.

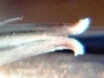


7916 H. mirabilis, Van Reenens Crest
7917 H. mirabilis, NE Dam VR Crest
7918 H. mirabilis,Van Reenens Crest, SE Dam
7959 H. mirabilis, W Van Reenens Crest
Subset 3. H. mirabilis – western zone, Greyton area
(cf. vars. beukmannii, rubrodentata).

7978 H. mirabilis, Schuitsberg
Subset 4. H. mutica – north-eastern zone, Buffeljags
(Swellendam area, cf. H. groenewaldii).




7801 H. mutica, Buffeljags. (3) 7889 H. mutica, Rotterdam 2.
Subset 5. H. floribunda – central zone, Swellendam area.


7774 H. floribunda, S Swellendam. 7963 H. floribunda, Niekerkshek.
Subset 6. H. mirabilis – outliers and ‘outgroups’.
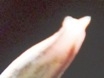



7248 H. mirabilis, Ballyfar, Infanta. (2) 7612 H. mirabilis, Diamant W. (2)



7780 H. retusa ‘geraldii’, Komserante
7781 H. retusa ‘foucheii’, Komserante
7974 H. mirabilis, Klipfontein, Potberg


7982 H. mirabilis. , Melkhoutkraal, Goukou
JDV87-132 H. parksiana, Mossel Bay
5. THE FLOWERS. Figures Set 5a and 5b
5a. Flower progression. Figures set 5a illustrate the ageing progression in two florets from two populations. One of the biggest hurdles in describing or illustrating the flower is the change that takes place that occurs in the life of the flower from bud opening to flower wilting over a period of 4-6 days. As can be seen in these successive images, the petal spread wider with age. But clearly there is a difference in basic structure of the individual flower that also determines attitude of the petals through the various stages. Colour and texture of the flower may also vary as the flower loses turgidity with aging and wilting.














5a.1 7913 H. mirabilis, Rietkuil















5a.2 7963 H. floribunda, Van Niekerkshek
Flower length in mm
| 5910 | 7918 | 7513 | 7262 |
| 14.3 | 13.5 | 11.8 | 14.5 |
| 13 | 13.5 | 12.1 | 13.7 |
| 12 | 13 | 12 | 14.6 |
| 12.5 | 13.4 | 12.5 | 14.6 |
| 14 | 13.1 | 13.4 | |
| 13 | 13.4 | 15 | |
| 13 | 13.2 | 15.3 | |
| 14 | 13.2 | 16.8 | |
| 12.5 | 13 | 16.7 | |
| 14.2 | 12 | 15.3 | |
| 12.5 | 12.8 | ||
| 13.5 | 12.5 | ||
| 12 | 12.4 | ||
| 14 | 12.6 | ||
| 13.5 | 12.5 | ||
| 12.3 | |||
| 12.5 | |||
| 12 |
Fig. 5a.3 Table – headed by collecting nos. Flower length in mm.
5b. Flowers. Figures Set 5. Flowers were photographed as available using fixed lens settings so that images were reasonable comparable. Some were taken and seized against a similarly photographed millimeter rule and these measurements are given in Figure 5a.3 above as a general guide to size and variation. This was only done for 4 sets of flowers because it is very evident that my original observation of variability of a high order is true. The measurement is simply of the maximum length of the flower from attachment to pedicel to furthest upper tip in four collections. Obviously this is not ideal as how the upper petals reflex (and with age too) will distort the result. But I used it, and submit it to provide a scale for any other measurement of those given flower that one would like to make. Smaller dimensions using a millimeter ruler are not remotely accurate enough over the error already present arising from method. The sizes given in species descriptions in the archaic way still practiced, are fairly useless as there is no indication of precision and any statistical measure of inherent deviation. The photographing method, despite changes also inherent in the picture editing process, allows reasonable size comparisons. The fact is that across the three subgenera, tube length is consistently in the area 8-10mm probably corresponding to a common pollinator. An important consideration is also that of the requirements of a sampling process in statistics where randomness is essential. The tendency in the field is to be selective and this generates the problem of bias that is almost unavoidable even when this is considered.
A point that must be stressed is the question of plant condition on whatever structures are used for study. Generally if a plant flowers at all, it can be assumed that the condition of the plant was good enough and the flower appearance would be true. This is an assumption carried into most studies of herbarium material and generally accepted as valid. But when decisions are based on the nature of whole inflorescences, it becomes quite evident that plants in different niches and habitats, and even different seasons, will produce inflorescences according to plant vigour. Over and above this is the variability that derives from habitat adaptation. Proliferating ecotypes on well-drained cliff-faces will produce quite a different inflorescence to that of solitary ecotypes in deeper moisture-holding soils. Flowers on an inflorescence lasting 3-4 days from initial petal spread to wilting also change with time. The position on the flower is also significant as the terminal flowers are invariably smaller. Another problem is the assumption that characters are independent. The fact is that, say, the bud-tip is flattened. Petals contained in that bud-tip are constrained to fold accordingly and if the petals vary in width or length they will display differently simply because of the containment within the bud-tip.
The populations listed in Table 1 were visited during February when they were in flower, and a series of flowers photographed in each population as available. Each flower was photographed in profile and from face view with the camera lens at a fixed focus setting. It is just a fact that all Haworthia flowers are closely the same in respect of size as stated above. I am aware of smaller flowers in H. wittebergensis, as one example, and also very large flowers to nearly 30mm long (against an average of approximately 15-20mm) in H. herbacea from Villiersdorp. The numerical measurements are variable and while there is no doubt that these could be found if sought, the intention here is to show that they are unlikely to help resolve the issues of species differences at the level that they are needed. This is certainly not without very large samples and precise measurement to accuracy of at least 0.1mm. Some observations were recorded as described above of peduncles, capsules and seeds. But only to indicate that they have been considered and also regarded and seen as variables that are unlikely to make any contribution by virtue of the degree of similarity they show even over the few species involved in the observations. What differences are discernible are highly unlikely to have any significance when one thinks that these have to be shown to exist consistently in very similar structures over many populations and many species.
The largest differences one would expect to see should be in the individual flowers (florets). Pictures were thus taken of the flowers in profile and also of the facial aspect. Size is a variable and Fig. 5b.1 shows three flowers from different clones in 7910. Fig. 5b.2 shows two flowers from the base and tip of a single inflorescence in 7980 – the upper floret half the size of the lowest. Fig. 5b.3 is a diagram to show profile and face of a flower (6651) to suggest axes along which a flower could be orientated to obtain proper points of reference for measurements of angles or size. The face and profile are of 6651, but the three part flower bases are all from the same collection. These virtually sum up the entire argument as the diagrams show a floret without a petiole, and various shapes to the base of the tube that I refer to as the perigon – the section before the petals separate. The taper from the pedicel onward (the stipa?) is very variable and may be abrupt or extended. The tube may be uniformly deep or constricted at the middle. A pronounced swelling (gaster) of the tube may occur below near the base. Other variables are the angle at which the flower stands to the peduncle. This one rather complicates the imaging of the face as I tried to photograph holding the camera at right angles to the peduncle and that does not properly capture a flower that is very vertical.
The sequence of illustrations onwards from 5b.4 is first the faces and then the profiles of the flowers according to the listing of the sets of populations in Table 1.
SET 5B.4 – FACES
Subset 1. H. mirabilis – Eastern Zone, Riversdale area.

6651 H. mirabilis, SW Riversdale
















7778 H. mirabilis, Komserante



































7818 H. mirabilis, Windsor































7809 H. mirabilis, Koeisekop
Subset 2. H. mirabilis – Central Zone, Swellendam area.



7887 H. mirabilis, Rotterdam










7912 H. mirabilis, Rietkuil











7913 H. mirabilis, Rietkuil











7916 H. mirabilis, Van Reenens Crest. 7917 H. mirabilis, Van Reenens Crest.

















7918 H. mirabilis, Van Reenens Crest
















7919 H. mirabilis, Van Reenens Crest







7955 H. mirabilis, Van Reenens Crest















7959 H. mirabilis, Van Reenens Crest





7960 H. mirabilis, Kruiskloof.
Subset 3. H. mirabilis – Western Zone, Greyton area.















7262 H. mirabilis, S Greyton


























7978 H. mirabilis, Schuitsberg






7979 H. mirabilis, Nethercourt












7980 H. mirabilis, E Ouplaas












7981 H. mirabilis, Schuitsberg N
Subset 4. H. mutica – Northeastern Zone, Buffeljags area.
















7801 H. mutica, Buffeljags









7888 H. mutica, Rotterdam 1









7889 H. mutica, Rotterdam 2
Subset 5 H. floribunda – Central Zone, Swellendam area.







7774 H. floribunda, S Swellendam















7910 H. floribunda, Rietkuil












7963 H. floribunda, Niekerkshek














7975 H. floribunda, Rotterdam 1, 7976 H. floribunda, Rotterdam
Subset 6. Outliers and ‘outgroups’.








7248 H. mirabilis, Ballyfar











7513 H. mirabilis, Klipfontein, Bromberg












7612 H. mirabilis, Diamant W





7780 H. retusa, Komserante. 7781 H. retusa, Komserante. 7915 H. mirabilis,Van Reenens Crest





7920 H. retusa ‘nigra’, Van Reenens Crest
7974 H. mirabilis (‘floribunda’), Klipfontein, Potberg
7982 H. mirabilis, Goukou




87-132 H. parksiana, Mossel Bay
7937 H. mutica, Spitzkop, Bredasdorp
SET 5B.4- PROFILES
Subset 1. H. mirabilis – Eastern Zone, Riversdale area.

6651 H. mirabilis, SW Riversdale
















7778 H. mirabilis, Komserante





































7818 H. mirabilis, Windsor


























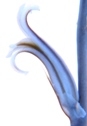




7809 H. mirabilis, Koeisekop.
Subset 2. H. mirabilis – Central Zone, Swellendam area.



7887 H. mirabilis, Rotterdam












7912 H. mirabilis, Rietkuil










7913 H. mirabilis, Rietkuil











7916 H. mirabilis, Van Reenens Crest. 7917 H. mirabilis, Van Reenens Crest.















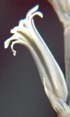

7918 H. mirabilis, Van Reenens Crest
















7919 H. mirabilis, Van Reenens Crest







7955 H. mirabilis, Van Reenens Crest














7959 H. mirabilis, Van Reenens Crest





7960 H. mirabilis, Kruiskloof
Subset 3. H. mirabilis – Western Zone, Greyton area.















7262 H. mirabilis, S Greyton



























7978 H. mirabilis, Schuitsberg






7979 H. mirabilis, Nethercourt












7980 H. mirabilis, E Ouplaas












7981 H. mirabilis, Schuitsberg N
Subset 4. H. mutica – Northeastern Zone, Buffeljags area.
















7801 H. mutica, Buffeljags









7888 H. mutica, Rotterdam 1










7888 H. mutica, Rotterdam 2
Subset 5 H. floribunda – Central Zone, Swellendam area







7774 H. floribunda, S Swellendam
















7910 H. floribunda, Rietkuil












7963 H. floribunda, Niekerkshek














7975 H. floribunda, Rotterdam 1. 7976 H. floribunda, Rotterdam.
Subset 6. Outliers and ‘outgroups’.







7248 H. mirabilis, Ballyfar











7513 H. mirabilis, Klipfontein, Bromberg












7612 H. mirabilis, Diamant W





7780 H. retusa ‘geraldii’, Komserante
7781 H. retusa ‘foucheii’, Komserante
7915 H. mirabilis, Van Reenens Crest





7920 H. retusa ‘nigra’, Van Reenens Crest
7974 H. mirabilis (‘floribunda’), S Klipfontein, Potberg
7982 H. mirabilis, Goukou


JDV87-132 H. parksiana, Mossel Bay
Haworthia flowers – some comments as a character source, part 1
Haworthia flowers – some comments as a character source, part 2
Haworthia flowers – some comments as a character source, part 3

Pingback: Mystery - part 9, S Tradouw - Haworthia Updates
Pingback: Haworthia flowers – some comments as a character source, part 3 | Haworthia Updates
Pingback: Haworthia flowers – some comments as a character source, part 1 | Haworthia UpdatesHaworthia Updates